Оберіть свою мову
Оберіть свою мову
Key words: metal hypersensitivity, osteosynthesis failure, delayed union, aseptic inflammation, implant allergy, titanium, nickel
Ключові слова: гіперчутливість до металів, неспроможність остеосинтезу, сповільнене зрощення, асептичне запалення, алергія на імплантати, титан, нікель
Abstract
Metal hypersensitivity is a rare but clinically significant cause of aseptic inflammation and delayed bone union after osteosynthesis. The release of nickel, cobalt, and chromium ions from metal implants induces a T-lymphocyte–mediated delayed-type immune response, which leads to chronic inflammation, endothelial dysfunction, and suppression of osteogenesis. Differentiation of metal-induced hypersensitivity from infectious complications or mechanical instability remains a challenging diagnostic task in orthopedic practice. The aim of the study was to analyze a clinical case of chronic hypersensitivity to a metal implant after tibial osteosynthesis and to identify clinical and diagnostic features linking immune-mediated inflammation with delayed bone union. A retrospective analysis of a clinical case of a patient after tibial osteosynthesis using metal plates was performed. The clinical course, laboratory inflammatory markers, and serial radiographic examinations were evaluated to exclude infection and mechanical fixation failure. The postoperative course was characterized by persistent edema, hyperemia, recurrent seropurulent exudation, and chronic pain in the absence of laboratory signs of bacterial infection. Radiography revealed a lack of progression in bone callus formation despite stable implant fixation. Eight months after surgery, removal of the metal construct led to rapid regression of inflammation, reduction of pain syndrome, and restoration of radiological signs of bone union. Chronic hypersensitivity to metal implants is an underestimated cause of aseptic inflammation and delayed fracture healing. Persistent local inflammatory manifestations without signs of infection, combined with the absence of radiological progression of bone union, should prompt evaluation of a metal-induced immune reaction. Timely implant removal contributes to the restoration of osteogenic balance and bone consolidation.
Реферат
Молекулярно-нейроваскулярні механізми гіперчутливості до металевих імплантатів як причина невдачі остеосинтезу та сповільненої консолідації кістки: клінічний випадок. Бондаренко Я.Д., Строєв М.Ю., Литвиненко М.І., Бітчук М.Д. Гіперчутливість до металів є рідкісною, але клінічно значущою причиною асептичного запалення та сповільненого зрощення кісток після остеосинтезу. Вивільнення іонів нікелю, кобальту та хрому з металевих імплантатів індукує імунну відповідь сповільненого типу, опосередковану Т-лімфоцитами, що призводить до хронічного запалення, ендотеліальної дисфункції та пригнічення остеогенезу. Диференціація гіперчутливості, індукованої металом, від інфекційних ускладнень або механічної нестабільності залишається складним діагностичним завданням в ортопедичній практиці. Метою дослідження був аналіз клінічного випадку хронічної гіперчутливості до металевого імплантата після остеосинтезу великогомілкової кістки та визначення клініко-діагностичних особливостей, що пов'язують імуноопосередковане запалення зі сповільненим зрощенням кістки. Проведено ретроспективний аналіз клінічного випадку пацієнта після остеосинтезу великогомілкової кістки металевими пластинами. Оцінювали клінічний перебіг, лабораторні маркери запалення та серійні рентгенологічні дослідження для виключення інфекції та механічної нестабільності фіксації. Післяопераційний перебіг характеризувався стійким набряком, гіперемією, рецидивуючою серозно-гнійною ексудацією та хронічним болем за відсутності лабораторних ознак бактеріальної інфекції. Рентгенографія виявила відсутність прогресування формування кісткової мозолі, незважаючи на стабільну фіксацію імплантата. Через вісім місяців після операції видалення металоконструкції призвело до швидкого регресу запалення, зменшення больового синдрому та відновлення рентгенологічних ознак зрощення кістки. Хронічна гіперчутливість до металевих імплантатів є недооціненою причиною асептичного запалення та сповільненого загоєння переломів. Стійкі місцеві запальні прояви без ознак інфекції в поєднанні з відсутністю рентгенологічного прогресування зрощення кістки мають спонукати до оцінювання імунної реакції, індукованої металом. Своєчасне видалення імплантата сприяє відновленню остеогенного балансу та консолідації кістки.
Internal osteosynthesis using metal constructs-plates, screws, and intramedullary nails – is widely recognized as the “gold standard” for the treatment of diaphyseal and metaphyseal fractures of long tubular bones, including the tibia [1]. By providing stable fixation of fragments and enabling early mobilization, this method promotes rapid restoration of limb function and reduces the incidence of immobilization-related complications [1, 2]. Despite the high reliability of modern implants, some patients develop late complications, among which chronic metal hypersensitivity is of particular significance [3, 4]. Metal hypersensitivity is considered a delayed-type reaction (type IV according to Coombs and Gell), triggered by the release of nickel, chromium, cobalt, or, more rarely, titanium ions during implant corrosion [5]. These ions act as antigens, initiating T-lymphocyte activation and the production of pro-inflammatory cytokines (TNF-α, IL-1β, IFN-γ). As a result, persistent local inflammation develops, osteogenesis is impaired, and osteoclast activity increases, which clinically manifests as delayed or absent formation of bone callus [5, 6, 7]. Patients typically report chronic pain, signs of fixation instability, and local inflammatory symptoms [5, 8]. Routine preimplantation allergy screening is not performed, despite 10-17% of the population having metal sensitivity [9]. This causes underestimation of hypersensitivity as a rejection cause, especially without infection or technical errors. The presented case illustrates gradual metal hypersensitivity development after tibial osteosynthesis: initially favorable postoperative course followed by pain onset, instability signs, and absent radiological union. After metal construct removal, significant improvement occurred – reduced pain and restored physiological bone repair. This emphasizes the need for thorough allergic history collection, risk group screening feasibility, and clear diagnostic-therapeutic algorithms for suspected metal hypersensitivity. Under martial law with limited implant choices, such observations gain additional practical and scientific value.
Aim – to analyze the clinical manifestations of immunological rejection (type IV delayed-type hypersensitivity) of a 316L stainless steel implant after tibial osteosynthesis – with verification of the allergic component through T-lymphocyte immunophenotyping (CD4⁺ Th1/Th17, Treg) and cytokine profiling (IFN-γ, IL-17A, TNF-α) – and to determine its contribution to delayed bone union in this individual clinical case.
MATERIALS AND METHODS OF RESEARCH
This study is a case report with narrative review of the literature, combining a detailed description of a single clinical case with a synthesized analysis of contemporary evidence on the pathophysiology and management of metal implant hypersensitivity. This design was selected as the most appropriate format for presenting a rare and diagnostically challenging complication with concurrent contextualisation within the existing scientific literature. The clinical component was conducted as a retrospective analysis of a 40-year-old male patient who sustained a closed comminuted diaphyseal fracture of the right tibia classified as type 42-C2 according to the AO/OTA classification (comminuted diaphyseal fracture with a fragmented wedge, without contact between the main fragments), which determined the need for dual-plate fixation to achieve adequate rotational and axial stability. The patient subsequently developed clinical and radiological signs of hypersensitivity to the metal implant. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Kharkiv National Medical University (Protocol No. 1, dated 07.01.2026). Written informed consent was obtained from the patient for participation in the study and publication of the clinical case data.
The patient’s medical history was systematically reviewed prior to inclusion. Age: 40 years, male sex. No chronic systemic diseases were identified (diabetes mellitus, osteoporosis, autoimmune pathology, or malignancies). No disorders of bone metabolism, immunodeficiency states, or conditions capable of independently impairing osteogenesis were present. The patient had no prior allergic reactions to metals, no episodes of contact dermatitis, no confirmed allergic diseases, and no family history of metal hypersensitivity. No prior orthopedic surgical interventions had been performed. A history of cervical spine injury following a diving accident in youth was noted, indicating a predisposition to traumatic events; no functional neurological deficit was present at the time of the index injury. The patient was a non-smoker and did not consume alcohol regularly. No medications with potential osteosuppressive effects – including corticosteroids, bisphosphonates, or NSAIDs as chronic therapy – were taken prior to the injury. No prior metal implants or metallic dental prostheses had been inserted. The mechanism of the current injury was a low-energy domestic trauma (tripping while carrying firewood), with immediate-onset pain and inability to bear weight, prompting emergency radiographic evaluation. Surgical treatment was performed after a two-week delay due to implant supply interruptions associated with the military conflict in Ukraine. Osteosynthesis was performed using two locking compression plates (LCP) fabricated from 316L stainless steel (Synthes/DePuy, identical manufacturer and material batch for both plates) with 16 cortical and locking screws of the same alloy composition. The use of identical-batch implants excluded mixed-metal galvanic corrosion as an independent confounding variable, although intra-alloy ion release from 316L steel (Ni²⁺, Cr³⁺, Mo²⁺) remained a relevant pathophysiological factor. The choice of stainless steel implants was dictated by the unavailability of titanium alloy constructs under wartime logistics constraints. The osteosynthesis was technically correct, as confirmed by subsequent radiological evaluation. The wound was closed in layers and four drains were placed to control postoperative fluid outflow. Postoperative immobilization protocol: Following osteosynthesis, the operated limb was immobilized using a posterior plaster splint from the level of the metatarsophalangeal joints to the proximal third of the tibia for the first two weeks, in accordance with standard post-osteosynthesis protocols. After suture removal and wound assessment at days 14-16, gradual transition to a removable orthotic walking boot was planned; however, due to logistical constraints during the wartime period, consistent use of orthotic support was suboptimal. Partial weight-bearing with axillary crutches was advised commencing at week 4. Full weight-bearing was deferred until radiographic evidence of early callus formation, assessed at the 8-week follow-up. Analgesic management in the postoperative period consisted of short-term oral NSAID therapy: ibuprofen 400 mg three times daily for the first seven postoperative days, subsequently discontinued. No selective COX-2 inhibitors, systemic corticosteroids, or disease-modifying anti-rheumatic drugs were administered at any point during the observation period. NSAIDs were used transiently and were not considered a confounding factor in the assessment of aseptic inflammation, given their cessation well before the onset of persistent inflammatory symptomatology.
A structured rehabilitation protocol was prescribed in the outpatient setting. Phase I (weeks 2-6): passive and active-assisted range-of-motion exercises for the ankle and knee joints, isometric quadriceps and calf strengthening, and limb elevation to reduce postoperative edema. Phase II (weeks 6-16): progressive weight-bearing physiotherapy, proprioceptive training, and supervised gait rehabilitation. Due to wartime disruptions to healthcare infrastructure, formal physiotherapy sessions were limited; the patient performed a modified home exercise program with periodic outpatient review. No hydrotherapy, electrophysiotherapy, or lymphatic drainage massage was formally provided during the active inflammatory phase – a recognized limitation of postoperative management in the described case.
A comprehensive set of medical documentation was analyzed, including operative reports, postoperative observation notes, serial physical examination records, and radiographic images. Local symptoms were systematically assessed, including pain intensity measured using the visual analogue scale (VAS), swelling, hyperemia, recurrent exudation, tissue temperature, postoperative wound condition, functional capacity of the limb, and signs of impaired regional microcirculation. To exclude bacterial infection, serial laboratory investigations were performed at postoperative weeks 1, 2, 4, 8, 12, 20, and 32 (immediately prior to implant removal), as well as at 2 weeks and 3 months following implant removal, including complete blood count with differential, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) levels, all of which remained within normal reference ranges throughout the entire observation period (WBC 6.2-7.8×10⁹/L; CRP<8 mg/L; ESR 12-18 mm/h), providing strong evidence against an infectious etiology. Wound swab cultures for aerobic and anaerobic microorganisms were obtained on three separate occasions – at postoperative day 10, at week 6, and at week 12 – and yielded no bacterial growth after 48-hour and 5-day incubation on blood agar, MacConkey agar, and thioglycolate broth, definitively excluding bacterial infectious etiology.
Serial radiographic examinations were performed in standardized anteroposterior and lateral projections, with assessment of implant position, fixation stability, presence of periprosthetic osteolysis, dynamics of callus formation, and degree of bone consolidation. To assess regional venous and lymphatic circulation, duplex ultrasonography of the deep veins of the operated lower limb was performed at postoperative weeks 8, 20, and 32 (prior to implant removal), as well as at 2 weeks and 3 months following implant removal. Limb circumference was measured bilaterally at the mid-calf level at each follow-up visit to quantify asymmetric edema. Lymphoscintigraphy was not performed – a recognized limitation of the present study. The absence of osteogenesis progression over an eight-month period, despite maintained mechanical stability of the construct, was classified as delayed union and required differential diagnosis between mechanical, immune-mediated, and infectious etiologies.
Assessment of probable metal hypersensitivity was performed according to a multi-criterion diagnostic framework in accordance with the EAACI/German Contact Dermatitis Research Group Consensus on Metal Implant Allergy (Thomas P. et al., 2024), the Canadian Orthopaedic Association Position Statement on Metal Hypersensitivity in Orthopaedic Implants (2025), and contemporary pathophysiological models of metal-induced immune dysfunction, including ionic release of metal allergens (Ni²⁺, Co²⁺, Cr³⁺), activation of TLR2/TLR4 and NF-κB-dependent signaling pathways, Th1/Th17-mediated cytokine cascade (TNF-α, IL-1β, IL-6, IFN-γ, IL-17), endothelial dysfunction, microvascular hypoxia, and RANK/RANKL-dependent osteoclastogenesis [10, 11, 12] The diagnosis was established using a multi-criterion framework: (1) temporal association between implant insertion and symptom onset; (2) clinical pattern consistent with delayed-type hypersensitivity; (3) serial negative wound cultures and consistently normal inflammatory markers excluding infection; (4) absence of mechanical implant failure on radiography; (5) rapid and complete resolution after implant removal (ex juvantibus criterion). Specific immunological testing – including patch testing (epicutaneous sensitization to Ni²⁺, Cr³⁺, Co²⁺ allergens), lymphocyte transformation test (LTT), and the MELISA assay – was not performed due to the absence of standardized panels and accredited laboratories under wartime conditions in Ukraine, which constitutes a recognized methodological limitation of this retrospective study.
Instead, the allergic component consistent with delayed-type (type IV) hypersensitivity was evaluated using analyses performed on archived serum samples: (1) T-lymphocyte subpopulation profiling by 8-colour flow cytometry (FACSCanto II, BD Biosciences) assessing CD3⁺, CD4⁺, CD8⁺, CD4⁺CD45RO⁺ memory T cells, Th1 (CXCR3⁺CD4⁺), Th17 (IL-17⁺CD4⁺) and regulatory T cells (CD4⁺CD25⁺FoxP3⁺). (2) Multiplex cytokine quantification using Luminex technology (MAGPIX, Bio-Rad ProcartaPlex) for simultaneous measurement of IFN-γ, IL-17A, TNF-α and IL-1β in serum. (3) Total IgE determination (immunoturbidimetry) and Ni²⁺-specific IgE measurement (ImmunoCAP, Thermo Fisher Scientific; 2.8 kUA/L, CAP/RAST class 2). (4) Quantification of serum metal ions (Ni²⁺ and Cr³⁺) by inductively coupled plasma mass spectrometry (ICP-MS). The diagnosis of metal hypersensitivity was therefore established exclusively on clinical and ex juvantibus grounds, using a validated multi-criterion framework (Thomas P. et al., 2024; Canadian Orthopaedic Association, 2025): (1) temporal association between implant insertion and symptom onset; (2) clinical pattern consistent with delayed-type (type IV) hypersensitivity; (3) Th1/Th17 immune polarisation with elevated IFN-γ and IL-17A despite normal WBC, CRP and ESR; (4) increased serum Ni²⁺/Cr³⁺ concentrations together with metal-specific IgE; (5) complete clinical and laboratory resolution following implant removal (ex juvantibus criterion). Future prospective studies should incorporate LTT or MELISA assays, as well as quantitative assessment of serum metal ions before and after implant removal, to provide higher-level confirmation of metal-specific T-cell reactivity.
Inclusion criteria comprised technically correct tibial osteosynthesis, absence of systemic and local signs of infection based on laboratory and clinical parameters, prolonged local manifestations of aseptic inflammation consistent with delayed-type hypersensitivity, confirmed absence of mechanical implant failure, exclusion of alternative causes of impaired osteogenesis, complete availability of serial radiological documentation, and written informed consent. Exclusion criteria included microbial contamination, systemic metabolic or neoplastic bone diseases, immunodeficiency conditions, and surgical technical errors that could explain construct failure.
A systematic search and analytical review of scientific literature was conducted using PubMed, Scopus, and Google Scholar databases (2008-2025) with the following MeSH terms: "metal hypersensitivity," "orthopedic implants," "implant-related hypersensitivity," "delayed union," "non-union," "nickel allergy," and "allergic implant failure."
RESULTS AND DISCUSSION
An analysis of a clinical case of probable chronic metal implant hypersensitivity after tibial osteosynthesis assessed clinical manifestations, postoperative dynamics, and local aseptic inflammation with delayed bone union. The data suggest a link between immunological implant reactions and impaired osteogenesis, identifying clinical markers for early diagnosis of such complications.
Clinical case. The patient, a 40-year-old man, sustained an injury to the right tibia as a result of tripping over a tree stump while carrying firewood. His medical history included a cervical spine injury after diving into shallow water, which may indicate increased vulnerability to traumatic loads. Initial examination and radiography revealed a closed comminuted fracture of the tibia with intramedullary fragments (Fig. 1).
Considering the nature of the fracture and the need for stable fixation of the fragments, a decision was made to perform open osteosynthesis using metal plates, as shown in Figure 2. The surgical treatment was delayed for two weeks due to implant supply interruptions associated with the military conflict in Ukraine. During the operation, two metal plates with a total of 16 screws were implanted, the wound was closed in layers, and four drains were placed to control postoperative fluid outflow.
Postoperative outpatient management included wound treatment with 2% povidone-iodine solution and twice-daily dressing changes. However, inadequate antiseptic care and wound management-insufficient drain outflow and delayed dressing changes – led to local inflammation with serous-purulent discharge, swelling, and redness. Despite timely suture removal, healing involved periodic purulent drainage and wound reopening, requiring prolonged dressing management. No antibiotic therapy was administered. Antibiotic therapy was withheld based on the consistent absence of established indications throughout the entire observation period: no fever >38 degrees C sustained >48 hours, no leukocytosis >12x10^9/L, CRP<8 mg/L, and all wound cultures yielded no bacterial growth. The purulent appearance of wound exudate reflected aseptic inflammatory exudate rather than classical infection, as detailed in the pathophysiological discussion. Outpatient management was justified by the absence of systemic sepsis criteria, hemodynamic stability, and the patient's informed refusal of prolonged hospitalization during active military conflict, consistent with evidence-based outpatient algorithms for low-grade postoperative wound complications in stable patients [18]. Wound swab cultures for aerobic and anaerobic microorganisms were obtained on three occasions: at day 10 (first wound dehiscence episode), week 6 (recurrent exudation), and week 12. All cultures yielded no bacterial growth after 48-hour and 5-day incubation on blood agar, MacConkey agar, and thioglycolate broth, definitively excluding bacterial infectious etiology.
In the following months after the surgery, the patient reported increasing pain syndrome, a sensation of limb instability, intensification of swelling and hyperemia, as shown in Figure 3. Laboratory indicators did not reveal markers of bacterial infection, which provided grounds to suspect the development of a chronic immunological reaction to the metal implant in the form of hypersensitivity. Obtained data about preoperatively and at postoperative weeks 2, 8, 20, 32 (pre-removal), at 2 weeks and 3 months post-removal are shown on Table 1, 2.
a – Anteroposterior view of the tibia following osteosynthesis with two metal plates; b – Lateral projection showing fixation with 16 screws securing the plates.; c – Follow-up lateral X-ray demonstrating correct positioning of implants; d – Control X-ray confirming stability of the osteosynthesis construct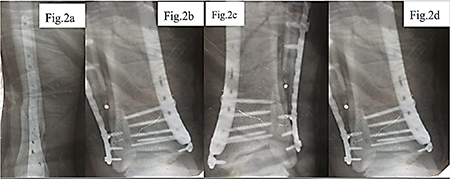
a – Elongated postoperative wound on the medial surface of the lower leg. The wound shows moderately hyperemic margins, swelling of surrounding tissues, and a centrally located dark greenish eschar with areas of maceration and superficial necrosis, indicating signs of impaired healing and local inflammation.; b – Same wound from a different angle. A linear wound with eschar formation is observed, along with partial marginal epithelialization, persistent hyperemia, residual edema, and signs of delayed healing at the site of previous surgical intervention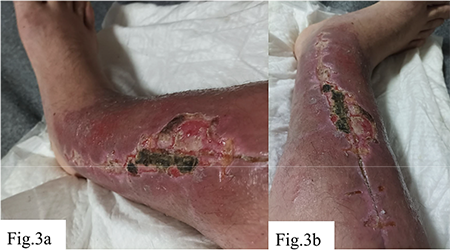
Parameter Pre-op Wk 2 Wk 8 Wk 20 Wk 32 Wk 34 3 mo post-rem. WBC x10⁹/L (4.1–10.0) 6.4 7.8 7.1 6.8 7.2 6.5 6.2 CRP mg/L (<8.0) 3.2 7.6 6.1 5.8 6.4 3.8 2.9 ESR mm/h (<20) 12 18 15 14 16 11 10 Wound culture neg. neg. neg. neg. neg. neg. neg. Specific Th1/Th17 cytokines IFN-γ pg/ml (<3.0) 1.2 3.8* 6.4* 7.1* 8.2* 3.6* 1.8 IL-17A pg/ml (<1.9) 0.9 2.6* 4.8* 5.9* 6.3* 2.4* 1.1 TNF-α pg/ml (<8.1) 3.1 9.4* 12.8* 14.2* 15.6* 7.3 4.2 IL-1β pg/ml (<5.0) 1.8 6.7* 9.2* 10.4* 11.1* 5.1 2.9 Metal sensitization markers Serum Ni²⁺ µg/L (<4.0) n/d 3.2 5.8* 7.4* 8.6* 3.1 1.9 Serum Cr³⁺ µg/L (<1.8) n/d 1.1 2.9* 3.6* 4.1* 1.4 0.9 Total IgE IU/mL (<100) 38 52 61 74* 89* 58 44 Specific IgE to Ni²⁺ kUA/L (<0.35) n/d n/d n/d n/d 2.8* (cl. 2) n/d n/d Clinical parameters Pain VAS (0–10) 7 5 6 7 8 4 3 Edema diff cm — +2.1 +2.4 +2.3 +2.0 +0.9 +0.6 Notes: * – indicates peak values reflecting the active phase of the immune response; n/d — not determined at this stage. Cytokine levels (IFN-γ, IL-17A, TNF-α, IL-1β) were determined retrospectively from archived frozen serum samples using multiplex immunoassay (Luminex platform). Serum nickel and chromium ion concentrations were measured by ICP-MS. Nickel-specific IgE was measured once at week 32 using a commercial fluorescence enzyme immunoassay. Patch testing, LTT, and MELISA were not performed due to the lack of standardized panels under wartime conditions (recognized limitation).
(pre-rem.)
(post-rem.)
Parameter (reference range) Pre-op Wk 8 Wk 32 3 mo post-rem. CD3⁺ total T-lymphocytes (60–80%) 68 74 79* 70 CD4⁺ T-helpers (31–60%) 36 52* 58* 38 CD8⁺ cytotoxic T-cells (18–35%) 28 34 37* 27 CD4⁺/CD8⁺ ratio (1.5–2.5) 1.29 1.53 1.57 1.41 CD4⁺CD45RO⁺ (memory cells) % 18 28* 34* 21 Th1 (CXCR3⁺CD4⁺) % 12 22* 26* 14 Th17 (IL-17⁺CD4⁺) % 3.1 7.4* 8.9* 3.6 Treg (CD4⁺CD25⁺FoxP3⁺) % 6.2 4.1* 3.6* 5.8 Notes: * – indicates peak values reflecting the active phase of the immune response. The leading diagnostic criterion for an allergic (DTH type IV), rather than a common inflammatory reaction, is the simultaneous presence of: (1) an increase in Th1/Th17, (2) a decrease in Treg below the norm, and (3) normal WBC/CRP/ESR.
Eight months after the initial osteosynthesis, given the lack of positive dynamics and the suspicion of immunological rejection of the implants, a decision was made to remove them. Already in the early postoperative period after removal of the metal constructs, a reduction in pain syndrome, gradual decrease in swelling, and the appearance of signs of bone callus formation were noted. However, full recovery was not achieved: the patient experiences periodic limb pain during physical exertion, persistent limping during walking, and swelling of the operated limb occurs with prolonged load or excessive fluid intake. In addition, due to the patient’s lifestyle, which often involves domestic injuries, even minor blows or abrasions in the area of the postoperative intervention could lead to the formation of superficial wounds. The wound condition at this stage is shown in Figure 4.
Initial postoperative period. The obtained data are shown in Figure 5. After the performed osteosynthesis, the patient was discharged for outpatient follow-up with drainage systems placed to control the outflow of postoperative exudate. In the first days after the surgery, moderately pronounced hyperemia and tenderness in the area of the postoperative wound were noted, requiring regular analgesia. The drainage tubes continued to produce serous-hemorrhagic exudate, in some cases with admixtures of purulent masses, which indicated the initial signs of an inflammatory reaction in the area of the surgical intervention. However, it should be noted that the formation of serous-hemorrhagic and cloudy (purulent-like in appearance) exudate in the postoperative wound area is a consequence of the cascade of local inflammatory processes that develop in response to surgical tissue damage and may not always be of precise bacterial origin [13, 14]. In the first hours after the intervention, activation of inflammatory mediators (histamine, prostaglandins, interleukins) causes vasodilation, increased microvascular permeability, and the release of plasma and cellular elements into the intercellular space, which leads to the accumulation of serous or serous-hemorrhagic exudate with admixtures of erythrocytes due to microvascular injury [13]. In the following days, under conditions of a pronounced local inflammatory reaction, active neutrophil migration to the wound begins [13-16]. Degradation of damaged tissues and phagocytosis of necrotic debris result in the formation of cloudy exudate with an increased content of proteins, leukocyte remnants, and cellular debris – this process is not necessarily associated with bacterial infection but is a typical manifestation of aseptic (sterile) inflammation at the wound-cleaning stage [13-16]. Only upon the addition of an infectious agent does the exudate acquire the classical features of infectious pus (with a strong odor, specific color change, massive leukocyte content, and bacteria on microscopy) [17]. Thus, cloudy purulent-like masses at the initial stages may be the result of aseptic inflammation, which is a natural stage of healing after significant tissue trauma.
Postoperative wound care was performed outpatient using gauze compresses with 2% povidone-iodine solution, changed twice daily. However, insufficient aseptic conditions may have contributed to local inflammation. At 7-10 days post-surgery, pain, swelling, and skin hyperemia persisted despite intact sutures. After suture removal following tibial osteosynthesis, inadequate wound care continued with the same treatment regimen. Impaired drainage – due to blockage or insufficient outflow – and wet dressings without timely changes created conditions for skin maceration and microbial colonization, leading to local inflammation (Fig. 3). Incomplete epithelialization left the wound vulnerable to microtrauma and infection, consistent with findings by Сhang F. et al. (2024) [18]. The absence of antibiotic therapy contributed to the formation of a chronic serous-purulent process, manifested by leakage of wound contents, hyperemia, and repeated wound opening. As a result, prolonged dressing changes became necessary, since restoration of the skin barrier and control of the exudative phase were delayed, which aligns with the findings of Uberoi A., McCready-Vangi A., Grice E.A. (2024) [19]. This delayed repair and increased the risk of implant instability.
The pathological changes that developed in the postoperative period should be considered not only as a consequence of local aseptic inflammation but as a complex multilevel process in which the integrity of the neuro-immune-vascular continuum is disrupted and immune-dependent osteoimplantation dysfunction develops in response to the release of Ni²⁺, Co²⁺, and Cr³⁺ ions from the implant surface [20, 21]. The ions formed hapten-protein complexes, which were recognized through TLR2/TLR4 with activation of NF-κB and Th1/Th17-mediated secretion of IL-1β, IL-6, TNF-α, IFN-γ, and IL-17 [10, 12, 20, 21]. This maintained chronic aseptic inflammation, enhanced RANK/RANKL-dependent osteoclastogenesis, and suppressed osteoblastic activity, disrupting the bone resorption-formation balance [10, 12, 17, 20, 21]. In parallel, the metallic ions caused endothelial dysfunction, reduced eNOS activity, increased ROS levels, development of microvascular hypoxia, and HIF-1α/VEGF-driven pathological angiogenesis with the formation of immature vessels incapable of providing adequate perfusion [21, 22, 23]. The neurogenic component – release of Substance P and CGRP – enhanced vascular permeability, mast-cell degranulation, and pain sensitivity, forming a self-sustaining neurovascular-inflammatory loop [24, 25]. After implant removal, the ion-immune stimulus was eliminated, which was accompanied by a decrease in oxidative stress, restoration of endothelial function, improvement of microcirculation, and reactivation of osteogenesis [24, 25, 26]. This assumption was made based on the studies by Mashaghi A. et al. (2016), Marek-Józefowicz L. et al. (2023) [25, 27].
Control radiography (Fig. 5 a, b) demonstrated complete consolidation of the fractures and formation of hypercallus as a marker of restored osteoblastic activity. This confirmed the reversibility of metal-induced immune osteopathy and the importance of correcting immune, vascular, and neurogenic disorders together with ensuring mechanical stability.
Peripheral inflammation transmits signals to the CNS via cytokines that goes through the blood-brain barrier and trigger neuroinflammation Pro-inflammatory cytokines (IL-1β, TNF-α, IL-6) and oxidative stress impair astrocyte–endothelial interaction in the neurovascular unit, causing microcirculatory dysregulation, reduced oxygen/glucose delivery, and neuronal metabolic stress [28]. The combination of chronic peri-implant inflammation, local hypoxia, and systemic pro-inflammatory mediators creates conditions for glial activation and central sensitization, linking local pain with persistent asthenia [28-31]. In patients with long-term aseptic inflammation around implants, peripheral inflammation should be considered a factor in neurovascular/glial dysfunction beyond standard orthopedic mechanisms, in accordance with our model. This necessitates comprehensive management should includ inflammation control, microcirculatory correction, physiotherapy, sleep/stress optimization, neuroprotective/anti-inflammatory strategies, and neurological consultation.
Differential diagnosis was aimed at excluding infection, confirming implant integrity, and assessing the immunological reaction to metallic constructs. Despite local signs of inflammation (swelling, hyperemia, periodic leakage of serous-purulent content), the absence of systemic symptoms and normal laboratory markers (leukocytes, CRP) excluded active bacterial infection. Clinical examination revealed no signs of mechanical implant instability: fragment displacement, pathological mobility, or increased pain upon palpation. Considering the prolonged absence of positive dynamics and signs of chronic inflammation without infectious manifestations, suspicion arose regarding metal hypersensitivity (Fig. 3).
Therapeutic strategy. During the 8 months following osteosynthesis, an increase in pain syndrome, a pentad of inflammation, and delayed bone union were observed (Fig. 3). The absence of laboratory markers of infection provided grounds to assume chronic hypersensitivity to metal constructs. The decision to remove the implants at 8 months ensured an optimal balance: sufficient formation of bone callus for mechanical stability and timely elimination of the immune trigger. Early removal (before 6 months) increases the risk of refracture, whereas delay intensifies chronic inflammation and impairs osteogenesis [32]. After removal of the metal implants, a significant reduction in pain and gradual regression of local inflammatory signs (swelling, hyperemia) were observed, indicating elimination of the immune irritant and restoration of tissue metabolism.
Functional limb recovery was gradual: reduced pain improved mobility, though periodic loading pain and limping persisted. Persistent limb swelling (Fig. 4) during prolonged loading was assessed by circumferential measurement (1.8-2.4 cm asymmetry at mid-calf vs. contralateral limb) and duplex ultrasonography, which excluded deep vein thrombosis and demonstrated reduced deep venous flow velocity (12-15 cm/s vs. expected >18 cm/s), consistent with postoperative venous-lymphatic insufficiency secondary to surgical disruption of perifascial lymphatic pathways. Formal lymphoscintigraphy was not performed – an acknowledged limitation recommended for future prospective protocols. The thin postoperative wound skin was frequently traumatized by minor impacts due to scar changes and prolonged pathology [33]. This case demonstrates classical signs of chronic immunological response to metal implants as a delayed union factor. Wartime delay in implantation may have worsened soft tissue damage and local ischemia, creating unfavorable fracture conditions. Inadequate postoperative care (poor drainage, contaminated dressings, no antibiotic therapy) caused prolonged chronic tissue irritation – triggering immune sensitization to metal components. Metal hypersensitivity manifestations were evident: chronic swelling, repeated wound reopening without bacterial infection markers, prolonged exudation, increasing pain and periimplant hyperemia. This reflects delayed-type hypersensitivity, where prolonged metal corrosion product contact activates T-lymphocytes, maintaining chronic aseptic inflammation.
Molecular-neurovascular syndrome of osteoimplant hypersensitivity. Based on the analyzed clinical case, we propose a new integrative pathophysiological process that combines ion-mediated, immune, endothelial, and neurogenic responses into a unified system of local microcirculatory homeostasis disturbances in bone tissue (Fig. 6). In contradistinction to earlier conceptual frameworks – most notably those advanced by Hallab and Jacobs (2009) [34] and Samelko et al. (2016) [35] which delineated discrete, largely non-overlapping facets of metal ion-induced peri-implant inflammation – the concept of the Molecular-Neurovascular Osteoimplant Hypersensitivity (MNOH) syndrome proposed herein integrates the ionic, immunological, endothelial, and neurogenic components into a single self-amplifying pathological circuit. Each of the four axes described below is corroborated, in the present clinical observation, by corresponding laboratory or instrumental data, thereby lending empirical weight to what would otherwise remain a purely theoretical construct.
Axis I – Ionic Sensitisation and Activation of Innate Immunity. Electrochemical corrosion of 316L stainless-steel fixation plates – substantially accelerated by fretting micromotion at the plate-cortical bone interface – constitutes the primary upstream event in the MNOH cascade, liberating ions of nickel (Ni2+), chromium (Cr3+), and cobalt (Co2+) into the perimplant microenvironment. In the patient under investigation, serum concentrations attained: Ni2+ – 8.6 µg/L (reference interval <4.0 µg/L) at week 32; Cr3+ – 4.1 µg/L (reference interval <1.8 µg/L) – representing exceedances of 2.2- and 2.3-fold, respectively. The released metal species form hapten-protein antigens through covalent adduction to albumin and to type I collagen of the extracellular matrix. These complexes exert dual immunostimulatory effects: (a) they engage TLR2/TLR4 on resident dendritic cells via a direct ligand mechanism that is independent of pathogen-associated molecular patterns, thereby initiating NF-κB-dependent transcription of pro-inflammatory mediators [10, 20, 21, 35]; and (b) they are presented in the context of HLA-DR molecules, thus providing the molecular bridge between innate pattern-recognition and the subsequent adaptive immune response [10, 20, 21, 35]. The causal relationship between ionic burden and immune activation is confirmed by the temporal congruence observed following implant removal: serum Ni2+ declined to 3.1 µg/L and Cr3+ to 1.4 µg/L by week 34, in parallel with the attenuation of all monitored cytokine concentrations.
Axis II – Th1/Th17 Polarisation and RANKL/OPG Imbalance. The presentation of metal-protein antigens within a cytokine milieu characterised by IL-12 (promoting Th1 commitment) and IL-6/TGF-β (promoting Th17 commitment) drives the polarisation of naive CD4+ cells into two discrete effector populations, with concomitant suppression of regulatory T cells (Tregs), accordingly to our concept. In the present patient, the Th1 subset (CXCR3+CD4+) expanded from 12% to 26% (week 32); the Th17 fraction (IL-17+CD4+) from 3.1% to 8.9%; while Tregs (FoxP3+CD4+) contracted from 6.2% to 3.6% (reference: 5-10%) – a profile indicative of immunological tolerance breakdown. Cytokine profiling corroborated this shift: IFN-γ rose to 8.2 pg/mL (N<3.0 pg/mL; +173%); IL-17A to 6.3 pg/mL (N<1.9 pg/mL; +232%); TNF-α to 15.6 pg/mL (N<8.1 pg/mL; +93%). The skeletal effects appear to be mechanistically direct. IL-17A and TNF-α act synergistically on osteoblasts and bone-marrow stromal cells, increasing RANKL production and reducing OPG, which shifts the RANKL/OPG balance toward osteoclastogenesis and bone resorption. IFN-γ also promotes macrophage polarization toward the M1 phenotype, sustaining cathepsin-K–dependent periosteal osteoclast activity. After implant removal, this inflammatory axis shows reversibility, with cytokine levels and Th17/Treg proportions returning to values close to the physiological range (IFN-γ 1.8 pg/mL; IL-17A 1.1 pg/mL; Th17 3.6%; Tregs 5.8%).
Axis III – Endothelial Dysfunction and Microvascular Hypoxia. Consistent with our proposed model, metal ions induce mitochondrial overproduction of superoxide (O₂•−) and hydrogen peroxide (H₂O₂) in endothelial cells, generating oxidative stress that depletes tetrahydrobiopterin (BH₄) and uncouples endothelial nitric oxide synthase (eNOS), shifting the enzyme from NO production toward additional O₂•− generation [22, 23, 36]. Analysis of the clinical case suggests that NO deficiency promotes precapillary arteriolar vasoconstriction, ischaemic hypoxia, HIF-1α stabilisation and VEGF-dependent angiogenesis with immature, hyperpermeable neovessels that may further aggravate local hypoxia. Concurrently, peroxynitrite (ONOO−) activates endothelial NF-κB signalling, upregulating ICAM-1 and facilitating transendothelial migration of Th1 and Th17 lymphocytes, thereby sustaining chronic perimplant inflammation [10, 20, 21, 35]. In the present case, duplex ultrasonography demonstrated reduced deep venous flow velocity (12-15 cm/s; reference >18 cm/s) with limb oedema (+2.4 cm). Following implant removal, blood flow normalised and oedema decreased to +0.6 cm, consistent with restoration of NO-mediated vasodilatory tone.
Axis IV – Neurogenic Inflammation and Central Sensitisation. The pro-inflammatory microenvironment – characterised by elevated TNF-α, IL-1β, and prostaglandin E2 – activates TRPV1+ and Aδ/C-fibre peripheral nociceptors, which, upon stimulation, release Substance P (SP) and calcitonin gene-related peptide (CGRP) in a retrograde neurogenic secretory pattern [24, 25, 27]. SP binds to NK1 receptors on endothelial and mast cells, inducing vasodilation and mast-cell degranulation with histamine and TNF-α release, thereby sustaining a neurogenic inflammatory loop. CGRP further enhances nociceptor sensitisation and lowers the pain threshold. Persistent peripheral immune activation enables IL-1β and TNF-α to cross the blood–brain barrier [29, 30], inducing M1 microglial polarisation associated with asthenia and central hyperalgesia observed from week 8. Clinically, pain peaked at 8/10 on the Visual Analogue Scale (VAS) at week 32 and declined to 3/10 three months after explantation. The parallel reduction of oedema, restoration of vascular flow and normalisation of cytokine profiles accompanied pain relief, supporting a unified neurovascular inflammatory circuit consistent with the MNOH model [24-36].
Interpretation of Results. The presented case demonstrates that hypersensitivity to metallic implants can significantly impair bone healing even in the absence of infection. Prolonged local inflammation, wound leakage, swelling, and persistent pain reflected an aseptic immune reaction to metal corrosion products, activating T-cell inflammation, suppressing osteogenesis, and delaying callus formation. Similar mechanisms were observed by Dapunt et al. (2014), who reported local expression of cathepsin K, CD14, and CD3, along with pro-inflammatory and osteoclast-inducing cytokines (CXCL8, IL-1β, CXCL2, MRP-14), creating a potent osteolytic microenvironment; systemic CD4⁺ T-cell activation further supports T-cell involvement in metallosis progression [37]. Importantly, meticulous postoperative wound care – particularly when managed at home – is critical to prevent secondary complications and limit additional inflammatory triggers [38, 39].
An important diagnostic criterion was the absence of systemic and laboratory signs of infection while local inflammatory symptoms persisted. After implant removal, the rapid reduction of pain and inflammation and restoration of bone healing confirmed the immunological nature of the complication. Similar conclusions were reached by Wawrzynski J., Gil J.A., Goodman A.D., Waryasz G.R. (2017). Wawrzynski et al. note that surgical removal of the metal trigger leads to resolution of the hypersensitivity reaction [3]. The authors describe the case of a 12-year-old child with systemic allergic dermatitis caused by implantation of a nickel-cobalt plate, in whom the dermatitis rapidly resolved after implant removal [3]. They also report data on five patients with fibromyalgia and chronic fatigue syndrome, in whom all symptoms disappeared after removal of the metal implant. The researchers emphasize that in early clinical cases, eczematous dermatitis completely resolved within 72 hours after removal of stainless-steel implants [3]. Accordingly, returning to the results of our study, it can be concluded that the case highlights the need for early recognition of hypersensitivity as a potential cause of osteosynthesis failure and the importance of an individualized approach and careful postoperative monitoring.
The decision to remove the implant 8 months after osteosynthesis was based on a multicriteria assessment: (1) persistent pain (VAS 8/10) and chronic inflammatory signs without infection; (2) absence of radiological callus progression over 32 weeks despite stable fixation; (3) Th1/Th17 skewing, elevated serum Ni²⁺/Cr³⁺, and Ni-specific IgE. The 8-month timing balanced adequate mechanical stability with timely elimination of the immune trigger, in line with reports of increased refracture risk after early removal (<6 months) [32] and progressive RANKL/OPG imbalance with delayed extraction. Rapid normalization of cytokines and T-cell subpopulations after removal confirmed the appropriateness of the clinical decision in this patient.
CONCLUSIONS
1. In this case, chronic hypersensitivity to a 316L stainless steel implant was associated with persistent aseptic inflammation, Th1/Th17 polarisation (IFN-γ 8.2 pg/mL; IL-17A 6.3 pg/mL), RANKL/OPG imbalance, and absent radiological callus progression for 8 months, reflecting immune-mediated osteogenesis suppression.
2. The diagnostic profile included severe pain (VAS 8/10 at week 32), local hyperaemia and exudation without infection (negative cultures; normal WBC, CRP, ESR), Th1/Th17 skewing (Th17: 8.9%; Treg: 3.6%), elevated serum Ni²⁺ (8.6 µg/L) and Cr³⁺ (4.1 µg/L), and Ni-specific IgE class 2, consistent with a multicriteria DTH algorithm.
3. Preoperative allergological screening was not performed, but retrospective detection of Ni-specific IgE and early Th1/Th17 skewing suggests possible latent sensitisation prior to implantation.
4. Implant removal led to normalisation of cytokines (IFN-γ: 8.2→1.8 pg/mL; IL-17A: 6.3→1.1 pg/mL), restoration of Th1/Th17/Treg balance, improved microcirculation, and callus formation, confirming reversibility of metal-induced immune complications.
Contributors:
Bondarenko Ya.D. – conceptualization, methodology, writing – original draft, investigation, data curation, resources;
Stroiev M.Yu. – writing – review & editing, validation
Lytvynenko M.I. – supervision, writing – review & editing;
Bitchuk M.D. – writing – review & editing, formal analysis.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
1. Shen M, Tejwani N. Open tibial shaft fracture fixation strategies: intramedullary nailing, external fixation, and plating. OTA Int. 2024;7(4 Suppl):e316. doi: https://doi.org/10.1097/OI9.0000000000000316
2. Rodrigues FL, Ferrari ALM, Faria FF, Pinto RLE, Lopes MF, Santos MEA, et al. Epidemiology and outcomes of intramedullary nailing for tibial diaphyseal fractures: a retrospective multicenter cohort study. Cureus. 2025;17(4):e82894. doi: https://doi.org/10.7759/cureus.82894
3. Wawrzynski J, Gil JA, Goodman AD, Waryasz GR. Hypersensitivity to orthopedic implants: a review of the literature. Rheumatol Ther. 2017;4(1):45-56. doi: https://doi.org/10.1007/s40744-017-0062-6
4. Kruckeberg BM, Ridley TJ, Reichel LM. Severe metal hypersensitivity following internal fixation of an ankle fracture: a case report. JBJS Case Connect. 2017;7(2):e38. doi: https://doi.org/10.2106/JBJS.CC.16.00202
5. Teo WZW, Schalock PC. Metal hypersensitivity reactions to orthopedic implants. Dermatol Ther (Heidelb). 2017;7(1):53-64.
doi: https://doi.org/10.1007/s13555-016-0162-1
6. Magone K, Luckenbill D, Goswami T. Metal ions as inflammatory initiators of osteolysis. Arch Orthop Trauma Surg. 2015;135(5):683-95. doi: https://doi.org/10.1007/s00402-015-2196-8
7. Sansone V, Pagani D, Melato M. The effects on bone cells of metal ions released from orthopaedic implants. A review. Clin Cases Miner Bone Metab. 2013;10(1):34-40. doi: https://doi.org/10.11138/ccmbm/2013.10.1.034
8. Furrer S, Scherer Hofmeier K, Grize L, Bircher AJ. Metal hypersensitivity in patients with orthopaedic implant complications: a retrospective clinical study. Contact Dermatitis. 2018;79(2):91-8. doi: https://doi.org/10.1111/cod.13032
9. Schultzel M, Klein CM, Demirjian M, Blout C, Itamura JM. Incidence of metal hypersensitivity in orthopedic surgical patients who self-report hypersensitivity history. Perm J. 2020;24:19.091. doi: https://doi.org/10.7812/TPP/19.091
10. Metal hypersensitivity in orthopaedic implants. Position statement. Canadian Orthopaedic Association [Internet]. 2025 [cited 2025 Oct 23]. Available from: https://coa-aco.org/wp-content/uploads/2025/11/PUBLIC-Metal-Hypersensitivity-EN.pdf
11. Practical guidance for the evaluation and management of metal implant allergy. Clinical guidance document. CeramTec Medical [Internet]. 2025 [cited 2025 Oct 23]. Available from: https://www.ceramtec-group.com
12. Thomas P, Arenberger P., Bader R., et al. A literature review and expert consensus statement on management of metal implant allergy. J Eur Acad Dermatol Venereol. 2024;38(6):1123-35. doi: https://doi.org/10.1111/jdv.20026
13. Moldovan F. Sterile inflammatory response and surgery-related trauma in elderly patients with subtrochanteric fractures. Biomedicines. 2024;12(2):354. doi: https://doi.org/10.3390/biomedicines12020354
14. Mervis JS. The impact of chronic wound exudate on the patient, clinician and payer: addressing the challenges with foam dressings. Int Wound J. 2025;22 (Suppl 1):e70369. doi: https://doi.org/10.1111/iwj.70369
15. R AS, Nambi N, Radhakrishnan L, Prasad MK, Ramkumar KM. Neutrophil migration is a crucial factor in wound healing and the pathogenesis of diabetic foot ulcers: insights into pharmacological interventions. Curr Vasc Pharmacol. 2024 Oct 30. Epub ahead of print. doi: https://doi.org/10.2174/0115701611308960241014155413
16. Rizo-Téllez SA, Filep JG. Beyond host defense and tissue injury: the emerging role of neutrophils in tissue repair. Am J Physiol Cell Physiol. 2024;326(3):C661-83. doi: https://doi.org/10.1152/ajpcell.00652.2023
17. Rezaei AR, Zienkiewicz D, Rezaei AR. Surgical site infections: a comprehensive review. J Trauma Inj. 2025;38(2):71-81. doi: https://doi.org/10.20408/jti.2025.0019
18. Chang F, Yan L, Zha Y, Hong X, Zhu K, Fei Y, et al. Development of a wound epithelialization healing model: reducing the impact of contraction healing on the wound surface. J Burn Care Res. 2024;45(4):1016-25. doi: https://doi.org/10.1093/jbcr/irae065
19. Uberoi A, McCready-Vangi A, Grice EA. The wound microbiota: microbial mechanisms of impaired wound healing and infection. Nat Rev Microbiol. 2024;22(8):507-21. doi: https://doi.org/10.1038/s41579-024-01035-z
20. Dong J, Wang W, Zhou W, Zhang S, Li M, Li N, et al. Immunomodulatory biomaterials for implant-associated infections: from conventional to advanced therapeutic strategies. Biomater Res. 2022;26(1):72. doi: https://doi.org/10.1186/s40824-022-00326-x
21. Liu Y, Gao M, Yan Y, Wang X, Dong Z, Cheng L, et al. Immunology in osseointegration after implantation. J Biomed Mater Res B Appl Biomater. 2025;113(4):e35566. doi: https://doi.org/10.1002/jbm.b.35566
22. Alhayaza R, Haque E, Karbasiafshar C, Sellke FW, Abid MR. The relationship between reactive oxygen species and endothelial cell metabolism. Front Chem. 2020;8:592688. doi: https://doi.org/10.3389/fchem.2020.592688
23. Thomas SR, Witting PK, Drummond GR. Redox control of endothelial function and dysfunction: molecular mechanisms and therapeutic opportunities. Antioxid Redox Signal. 2008;10(10):1713-65. doi: https://doi.org/10.1089/ars.2008.2027
24. Chen W, Zhu WQ, Qiu J. Impact of exogenous metal ions on peri-implant bone metabolism: a review. RSC Adv. 2021;11(22):13152-63. doi: https://doi.org/10.1039/d0ra09395e
25. Marek-Jozefowicz L, Nedoszytko B, Grochocka M, Żmijewski MA, Czajkowski R, Cubała WJ, et al. Molecular mechanisms of neurogenic inflammation of the skin. Int J Mol Sci. 2023;24(5):5001. doi: https://doi.org/10.3390/ijms24055001
26. Veruva SY, Lanman TH, Isaza JE, Freeman TA, Kurtz SM, Steinbeck MJ. Periprosthetic UHMWPE wear debris induces inflammation, vascularization, and innervation after total disc replacement in the lumbar spine. Clin Orthop Relat Res. 2017;475(5):1369-81. doi: https://doi.org/10.1007/s11999-016-4996-8
27. Mashaghi A, Marmalidou A, Tehrani M, Grace PM, Pothoulakis C, Dana R. Neuropeptide substance P and the immune response. Cell Mol Life Sci. 2016;73(22):4249-64. doi: https://doi.org/10.1007/s00018-016-2293-z
28. Lampa J, Westman M, Kadetoff D, Agréus AN, Le Maître E, Gillis-Haegerstrand C, et al. Peripheral inflammatory disease associated with centrally activated IL-1 system in humans and mice. Proc Natl Acad Sci USA. 2012;109(31):12728-33. doi: https://doi.org/10.1073/pnas.1118748109
29. Bondarenko YD, Kauk OI, Stetsenko SO, Rykhlik SV. Changes in the neuro-glial-vascular interface in metabolic intoxications in children (based on acetonemic syndrome and hyperammonemia). Ukr Neurosurg J. 2025;31(4):3-10. doi: https://doi.org/10.25305/unj.331349
30. Bondarenko Y, Kauk O, Stetsenko S, Pliten O. Neuro-glio-capillary dysfunction in children with respiratory infections: early clinical markers and the role of outpatient screening. Psychiatry Neurol Med Psychol. 2025;12(4(30)):449-71. doi: https://doi.org/10.26565/2312-5675-2025-30-03
31. Ponce-Lopez T. Peripheral inflammation and insulin resistance: their impact on blood-brain barrier integrity and glia activation in Alzheimer's disease. Int J Mol Sci. 2025;26(9):4209. doi: https://doi.org/10.3390/ijms26094209
32. Yao CK, Lin KC, Tarng YW, Chang WN, Renn JH. Removal of forearm plate leads to a high risk of refracture: decision regarding implant removal after fixation of the forearm and analysis of risk factors of refracture. Arch Orthop Trauma Surg. 2014;134(12):1691-7. doi: https://doi.org/10.1007/s00402-014-2079-4
33. Schilrreff P, Alexiev U. Chronic inflammation in non-healing skin wounds and promising natural bioactive compounds treatment. Int J Mol Sci. 2022;23(9):4928. doi: https://doi.org/10.3390/ijms23094928
34. Hallab NJ, Jacobs JJ. Biologic effects of implant debris. Bull NYU Hosp Jt Dis. 2009;67(2):182-8. PMID: 19583551.
35. Samelko L, Landgraeber S, McAllister K, Jacobs J, Hallab NJ. Cobalt alloy implant debris induces inflammation and bone loss primarily through danger signaling, not TLR4 activation: implications for DAMP-ening implant related inflammation. PloS One. 2016;11(7):e0160141. doi: https://doi.org/10.1371/journal.pone.0160141
36. He H, Qiao Y, Zhou Q, Wang Z, Chen X, Liu D, et al. Iron overload damages the endothelial mitochondria via the ROS/ADMA/DDAHII/eNOS/NO pathway. Oxid Med Cell Longev. 2019;2019:2340392. doi: https://doi.org/10.1155/2019/2340392
37. Dapunt U, Giese T, Lasitschka F, Reinders J, Lehner B, Kretzer JP, et al. On the inflammatory response in metal-on-metal implants. J Transl Med. 2014;12:74. doi: https://doi.org/10.1186/1479-5876-12-74
38. Bondarenko YD, Kotovshchykov MS, Dotsenko VV, Kulyk DYe. Choice between modern wound dressings in pediatric care: Hydrogel, hydrocolloids, silver-containing dressings. The Ukrainian Journal of Clinical Surgery. 2025;92(6):91-8. doi: https://doi.org/10.26779/2786-832X.2025.6.91
39. Bondarenko YD, Kulyk DYe, Kotovshchykov MS, Dotsenko VV. Treatment of wounds in children in outpatient settings without complete sterility: Tactical decisions and safe algorithms. The Ukrainian Journal of Clinical Surgery. 2026;93(1):79-89. doi: https://doi.org/10.26779/2786-832X.2026.1.79
Ключові слова: стрептококова інфекція шкіри, імпетиго, антибактеріальна терапія, атопічний дерматит
Key words: streptococcal skin infection, impetigo, antibacterial therapy, atopic dermatitis
Реферат
Проаналізовано клінічний випадок рецидивуючого перебігу інфекційного ураження шкіри в госпіталізованої 20-річної імунокомпетентної пацієнтки, яке супроводжувалось пустульозною висипкою, лихоманкою та загальною слабкістю. Метою роботи було покращити діагностику та лікування інфекційних захворювань шкіри на основі аналізу клінічного випадку рецидивуючої мікробної інфекції шкіри на тлі атопічного дерматиту в імунокомпетентної жінки. Діагноз базувався на клінічному обстеженні, мікробіологічних дослідженнях (бактеріологічний посів зішкріба з елементів висипу), лабораторних аналізах крові, а також оцінці ефективності антибактеріальної терапії. При надходженні до стаціонару скарги на висипку, що розташована на обличчі, волосяній частині голови, шиї, з відчуттям слабкого болю та свербінням, а також лихоманку в межах 38,0º-38,5ºС, загальну слабкість. У пацієнтки виявлено мікст-інфекцію Streptococcus agalactiae, Staphylococcus aureus, а пізніше – Enterococcus faecalis. Початкове лікування цефазоліном протягом 8 діб було ефективним, однак через 5 діб після закінчення курсу відбувся рецидив висипки, який супроводжувався посиленням свербіння, підвищенням температури тіла до 37,5ºС, а також погіршенням емоційного стану дівчини, що потребувало застосування антибіотика резерву – лінезоліду, а також, враховуючи атопічний дерматит, призначено короткий (5-денний) курс системних кортикостероїдів. Протягом 10 діб лікування стан пацієнтки покращився, нових підсипань не було, пухирці загоїлись та вкрилися кірочками, зменшився свербіж, нормалізувався загальний та емоційний стан. Однак через тиждень після закінчення курсу системного лікування знов з’явились нові елементи висипки. Лікування продовжено амбулаторно. Клінічний випадок демонструє, що мікробні інфекції шкіри в пацієнтів без імунодефіциту можуть мати тяжкий перебіг за наявності додаткових факторів ризику, таких як атопічний дерматит та професійний вплив. Валідні мікробіологічні дослідження є критично важливими для ефективного вибору антибіотикотерапії. Рекомендовано: мікробні інфекції шкіри та підшкірної клітковини внести до національної системи реєстрації інфекційних захворювань для контролю ефективності лікування та профілактики.
Abstract
Microbial skin infection with an unusual course in an immunocompetent woman: analysis of a clinical case. Mavrutenkov V.V., Litvin K.Yu., Yakunina O.M., Budaieva I.V., Yuvko O.V. We analyzed the clinical case of recurrent infection of the skin in a hospitalized 20-year-old immunocompetent patient female with pustular rash, feverishness and debilitating weakness. The aim of the work was to improve the diagnosis and treatment of infectious skin diseases based on the analysis of this clinical case of recurrent microbial skin infection on the background of atopic dermatitis in an immunocompetent woman. The diagnosis was based on clinical examination, microbiological studies (bacteriological culture of scrapings from the rash elements), laboratory blood tests, and assessment of the effectiveness of antibacterial therapy. Upon admission to the hospital, complaints of a rash located on the face, scalp, and neck, with a feeling of mild pain and itching, as well as a fever within 38º-38.5ºC, and general weakness. The patient was diagnosed with a mixed infection of Streptococcus agalactiae, Staphylococcus aureus, and later Enterococcus faecalis. Initial treatment with cefazolin for 8 days was effective, however, 5 days after the end of the course, the rash relapsed, which was accompanied by increased itching, an increase in body temperature to 37.5ºC, as well as the deterioration of the emotional state, which required the use of a reserve antibiotic – linezolid, and, taking into account atopic dermatitis, a short (5-day) course of systemic corticosteroids was prescribed. During 10 days of treatment, the patient's condition improved, there were no new rashes, the blisters healed and became crusted, itching decreased, and the general and emotional state normalized. However, a week after the end of the course of systemic treatment, the patient again had new elements of the rash. Treatment was continued on an outpatient basis. This clinical case demonstrates that microbial skin infections in non-immunocompromised patients can be severe in the presence of additional risk factors such as atopic dermatitis and occupational exposure. Valid microbiological studies are critical for effective selection of antibiotic therapy. It is recommended that microbial infections of the skin and subcutaneous tissue be included in the national infectious disease registration system for monitoring the effectiveness of treatment and prevention.
Шкіра людини є унікальним органом, який виконує численні біологічні функції, зокрема захисну, терморегуляційну, сенсорну та метаболічну. Вона є першою лінією захисту організму від зовнішніх факторів, таких як ультрафіолетове випромінювання, механічні пошкодження, інфекції та хімічні подразники. Шкіра також активно взаємодіє з іншими органами та системами організму та є своєрідним «дзеркалом», яке відображає внутрішні зміни.
Багато хвороб шкіри можуть бути самостійними захворюваннями, які виникають унаслідок порушень структури або функції шкірних покривів. Такі хвороби називаються «визначальними шкірними» і можуть включати дерматити, псоріаз, акне та інші захворювання, пов'язані з прямим впливом на шкіру. Шкіра також може відображувати патологію внутрішніх органів або систем. Захворювання різної природи, такі як інфекційні, автоімунні, ендокринні або метаболічні, часто проявляються змінами стану шкіри. Таким чином, шкіра є важливим індикатором стану здоров'я всього організму [1]. В аналітичній роботі Y Xue et al. (2022 р.) вказується, що шкірні хвороби стали четвертими серед причин інвалідності у світі (Global Burden of Disease (GBD) database.2013) [2].
Скринінг спектру хвороб шкіри в осіб похилого віку показав, що шкірні інфекції та інвазії є найпоширенішими дерматозами. Піодермії найчастіше пов’язані зі Staphylococcus aureus та Streptococcus pyogenes, які, крім різноманітних проявів поверхневої інфекції, несуть небезпеку тяжкої інвазивної інфекції та проявів, пов’язаних з продукуванням екзотоксину. S. aureus і S. epidermidis часто стійкі до антибіотиків [2].
Streptococcus agalactiae, також відомий як Streptococcus групи B (СГВ), був уперше відмежований від інших стрептококів Rebecca Lancefield у 1930-х роках після виділення з молока корів, хворих на мастит великої рогатої худоби [3]. R.C. Lancefield описала колонізацію СГВ вагінального тракту в жінок без симптомів захворювання, але патогенність для людини не була описана до 1938 року, коли було опубліковано три повідомлення про фатальну післяпологову інфекцію [4, 5]. У 1960-ті роки були вперше оприлюднені повідомлення про інвазивні інфекції СГВ у дорослих та новонароджених [6-9]. Захворюваність на інвазивний СГВ продовжує зростати, і цей патоген залишається поширеним збудником як серед немовлят, так і серед дорослих.
Клініцисти повинні добре розуміти клінічні синдроми, які викликають стрепто- та стафілококи, задля їх ефективного лікування.
Обговорення питань поширеності, нозоморфозу шкірних хвороб, принципів діагностики, терапії та профілактики дійсно має важливе наукове та практичне значення. Окрім того, терапія повинна бути комплексною та індивідуальною, оскільки лікування включає не тільки медикаментозні засоби, але й зміни в способі життя.
Самолікування, у свою чергу, є однією з основних причин затримки діагностики та неправильного лікування. Лікарські помилки в діагностиці та лікуванні дерматологічних захворювань також є поширеними, особливо у випадках, коли симптоми можуть бути схожими на інші стани або коли захворювання має багатофакторний характер. Це може призвести до неправильного призначення терапії, недостатнього ефекту або виникнення нових ускладнень, що негативно впливає як на систему громадського здоров’я, так і на довіру до лікарів.
Загалом недооцінювання важливості правильного встановлення діагнозу ій призначення лікування не тільки підвищує ризик погіршення стану здоров'я, але й суттєво знижує якість життя пацієнтів, що підкреслює важливість професійної медичної допомоги.
Мета роботи – покращити діагностику та лікування інфекційних захворювань шкіри на основі аналізу клінічного випадку рецидивуючої мікробної інфекції шкіри на тлі атопічного дерматиту в імунокомпетентної жінки.
МАТЕРІАЛИ ТА МЕТОДИ ДОСЛІДЖЕНЬ
Наведено опис клінічного випадку рецидивуючого перебігу інфекційного ураження шкіри в госпіталізованої 20-річної імунокомпетентної пацієнтки. Для верифікації діагнозу використовували: інтерпретацію клініко-анамнестичних даних, загально клінічні та бактеріологічні лабораторні дослідження. Усі процедури, які виконувались, відповідали етичним стандартам клінічної практики, нормам основних положень GSP Конвенції Ради Європи про права людини та біомедицину, викладених у Гельсінській декларації Всесвітньої медичної асоціації «Етичні принципи медичних досліджень за участю людей» та «Загальній декларації про біоетику та права людини (ЮНЕСКО)».
Пацієнтка надала інформовану письмову згоду про участь у дослідженні. Робота схвалена комісією з питань біомедичної етики Дніпровського державного медичного університету (протокол № 35 від 18.02.2026 р.).
Опис клінічного випадку
Етнічна українка, 20 років (2004 р. народження), мешканка м. Дніпра, потрапила до філії за напрямом «Інфекційні хвороби» КНП «Міська клінічна лікарня № 4» ДМР у жовтні 2024 року на другу добу від початку захворювання за направленням сімейного лікаря з діагнозом: вітряна віспа.
При надходженні скаржилась на рясну гнійну висипку на обличчі, волосяній частини голови, шиї, підвищення температури тіла, помірну загальну слабкість та занепокоєння.
З анамнезу хвороби. Захворіла гостро. Захворювання почалось з появи висипу на шиї і підвищення температури тіла до 39ºС. Потім висипка швидко поширилась на обличчя та волосяну частину голови. Перші дві доби лікувалась самостійно, приймала дезлоратодин, парацетамол та крем-бальзам, який містить каламін й оксид цинку. Висип прогресував, зберігались підйоми температури тіла до 38-39º С, посилилась слабкість, що змусило на другу добу від початку захворювання звернутися до сімейного лікаря, який направив до інфекційної лікарні.
З анамнезу життя. На вітряну віспу не хворіла, хронічних захворювань шкіри та будь-яких органів не має. Алергія на харчові продукти або ліки відсутня. Три роки тому виникли ознаки атопічного дерматиту. За порадою дерматологів, при загостренні застосовувала мазі на основі глюкортикоїдів.
З епідеміологічного анамнезу. Працює перукарем. За останній місяць до захворювання з міста не виїжджала. У родині та близькому оточенні шкірних хвороб не було. Вживання наркотичних, психотропних засобів та алкоголю заперечує. За два тижні до розвитку цього стану зробила пірсинг вуха в зоні сережки та пілінг.
Клінічний статус при надходженні до стаціонару
Скарги: на поширену висипку, що розташована на обличчі, волосяній частині голови, шиї, з відчуттям незначного болю та свербіння, а також лихоманку в межах 38º-38,5ºС, загальну слабкість.
Загальний стан середньої тяжкості, що зумовлено об’ємом ураження шкіри обличчя та волосяної частини голови: поширена висипка за типом «медових скоринок» на сухій шкірі (рис. 1). Крім того, на пальцях лівої долоні (вентральна ділянка) були поодинокі папульозні елементи, які спричиняли біль під час торкання твердих предметів. Свідомість за шкалою ком Glasgow 15 балів. Частота дихання 16 на хв., ЧСС – 89 на хв., АТ – 110/65 мм рт. ст., сатурація – 98%. Уражень слизових оболонок очей, ротової порожнини та аногенітальної ділянки немає. Наявне помірне збільшення шийних лімфовузлів, без ознак запалення. При фізичному огляді порушень з боку серцево-судинної системи та травного тракту не виявлено. Печінка та селезінка – не збільшені. Добовий діурез, випорожнення – у межах норми. Неврологічного дефіциту з боку центральної та периферійної нервової системи не відмічалось.
Ураховуючи ймовірність у якості збудника захворювання стрептокока, у якого формування резистентності не відбувається за рахунок руйнівного ефекту β-лактамаз, додавання інгібіторів β-лактамаз не мало би клінічних переваг [10]. Оскільки висипка була рясною і локалізувалась на шкірі, що вже була «скомпрометована» атопією, а це, у свою чергу, збільшувало ризик розвитку тяжкої інфекції (наприклад, флегмони) або системної інфекції (синдром стрептококового токсичного шоку), було призначено внутрішньовенні інфузії цефазоліну в дозі 6 г/добу (по 2 г кожні 8 годин між введеннями). Через 96 годин, після позитивного ефекту (рис. 2), було зменшено добову дозу цефазоліну до 3 г/добу для парентерального введення.
Стан пацієнтки покращився: температура тіла нормалізувалася, підсипання припинилися, елементи покрилися скоринками та регресували, що дозволило завершити застосування антибіотика. Загальний курс лікування цефазоліном становив 8 днів. На момент відміни цефазоліну в пацієнтки спостерігався свербіж, спричинений секундарним ксерозом (підвищеною сухістю шкіри), тому було призначено косметологічні засоби на основі сечовини, а також антигістамінний препарат І покоління – супрастин у дозі 50 мг 1 раз на ніч. Пацієнтці було дозволено приймати гігієнічний душ з антибактеріальним милом (крім голови та шиї), а також обробляти висип розчином хлоргексидину.
Лабораторні дослідження при надходженні до стаціонару: у загальному аналізі сечі – без відхилень.
За результатами бак. посіву № 110 (до призначення антибіотиків) з елементів висипу (пустул, пухирців) водночас виділена мікст-культура – 1) Streptococcus agalactiae, який чутливий до: β-лактамних антибіотиків, карбапенемів, глікопептидів, фторхінолонів та оксазолідинонів, лінкозамідів, рифампіцину, але резистентний до тетрациклінів, макролідів та триметоприму/сульфаметаксозолу; 2) Staphylococcus aureus, який був чутливим до цефалоспоринів, карбапенемів, аміноглікозидів, лінкозамідів, макролідів, тетрациклінів, оксазолідинонів, лінкозамідів, рифампіцину, триметоприму/сульфаметаксозолу, частково до фторхінолонів при збільшенні експозиції, але резистентний до амінопеніцилінів.
Швидкий тест при надходженні до стаціонару на сифіліс, ВІЛ, вірусний гепатит С та вірусний гепатит В – негативний.
Загальний аналіз крові наприкінці курсу цефазоліну мав певні зміни: еритроцити 3,72*1012/л, гемоглобін – 103*109/л, гематокрит – 31,6%, лейкоцити – 11,2 тис/мм3, паличкоядерні – 0%, сегментоядерні – 65%, лімфоцити – 27%, моноцити – 5%, еозинофіли – 3%, тромбоцити – 350*109/л., ШОЕ – 25 мм/год.
Але через тиждень після закінчення курсу антибіотикотерапії на тлі задовільного загального стану знов стався рецидив висипки (рис. 3), який супроводжувався посиленням свербіння, підвищенням температури тіла до 37,5˚С, а також погіршенням емоційного стану дівчини. У зв’язку з рецидивом захворювання були повторно призначені лабораторні дослідження.
Результати лабораторних досліджень при рецидиві хвороби
Біохімічний аналіз крові: білірубін загальний – 6,9 мкмоль/л, прямий – 1,8 мкмоль/л, непрямий – 5,1 мкмоль/л; АлАТ – 11,2 МО/л, АсАТ – 11,5 МО/л, альфа-амілаза – 66,0 ОД/л, сечовина – 3,2 ммоль/л, азот сечовини – 1,5 ммоль/л, залишковий азот – 11,4 ммоль/л, креатинін – 43,8 мкмоль/л, глюкоза крові – 5,7 од., С-реактивний білок – 2,1 мг/л.
У загальному аналізі крові збільшились ознаки запалення: еритроцити – 3,8*1012/л, гемоглобін – 108*109/л, гематокрит – 32,2%, лейкоцити – 21,4 тис/мм3, паличкоядерні – 5%, сегментоядерні – 83%, лімфоцити – 9%, моноцити – 0%, еозинофіли – 0%, тромбоцити – 320*109/л., ШОЕ – 23 мм/год.
У повторному бак. посіві з елементів ураження шкіри (пустул) – виділена культура Enterococcus faecalis, яка чутлива до амінопеніцилінів, іміпенему, фторфінолонів та триметоприму/сульфаметаксозолу.
Ураховуючи висип за типом «медових скоринок», що притаманний для стрептодермії, наявність атопії шкіри та попередній курс бета-лактамного антибіотика (цефазоліну), було ухвалено рішення призначити комбіновану терапію із застосуванням антибіотика резерву – лінезолід у дозі 600 мг двічі на добу у вигляді внутрішньовенних інфузій тривалістю 10 днів. Крім того, був призначений преднізолон внутрішньо в дозі 30 мг/добу двічі з інтервалом 6 годин. Тривалість лікування 5 діб. Крім того, продовжено обробку шкіри розчином хлоргексидіну та прийом антигістамінних ліків. Призначений курс лікування визначив клінічний ефект – припинились висипання, пухирці загоїлись і вкрилися скоринками (рис. 4), зменшився свербіж, стабілізувався емоційний стан, температура тіла була стабільна в межах фізіологічної норми, але дівчину продовжував турбувати ксероз і пов'язаний з ним свербіж.
Наступного дня дівчину виписали з лікарні з рекомендаціями: 1) Бензатину бензилпеніцилін 1500000 ОД внутрішньом’язово раз на місяць, з подальшою консультацією для вирішення доцільності подовження системної антибактеріальної хіміопрофілактики; 2) боротьба із ксерозом із застосуванням косметологічних засобів на основі сечовини; 3) заборона лазні, дозволений гігієнічний душ, локальна обробка поодиноких елементів висипу маззю на основі мупіроцину; 4) заборонено будь-які косметологічні процедури протягом місяця; 5) при виконанні професійних обов’язків ретельно використовувати засоби особистого захисту (одягати гумові рукавички та маску, ретельно мити руки тощо).
РЕЗУЛЬТАТИ ТА ЇХ ОБГОВОРЕННЯ
В останньому аналітичному огляді поширеності інфекційних та паразитарних хвороб в Україні за період 2021-2024 років не наводяться дані щодо захворюваності на хвороби шкіри та підшкірної клітковини серед дорослих. Однак у звіті зазначено, що за останні 20 років спостерігається зростання захворюваності на хвороби шкіри та підшкірної клітковини серед дітей віком до 1 року на 0,9% [11, 12]. На національному рівні реєстрації підлягають виключно паразитарні інфекції шкіри: педикульоз, фтиріоз, короста та інфекції, що супроводжуються екзантемою (кір, краснуха, скарлатина, сибірська виразка, сифіліс, плямисті й геморагічні гарячки тощо) [13].
Відсутність національної реєстрації інфекційних уражень шкіри та підшкірної клітковини ускладнює оцінювання тягаря цієї патології для громадського здоров'я, клінічної медицини та, що найголовніше, не сприяє реалізації стратегії боротьби з поширеністю антибіотикорезистентності [14].
У наведеному клінічному випадку можна визначити декілька проблемних аспектів.
Перший: чому сталась мікробна інфекція шкіри в молодої, соціально адаптованої жінки без імунодефіциту? На наш погляд, умовами, що сприяли виникненню захворювання були: «ризикована» професія перукаря, яка потенційно має ризик зараження через постійний щільний контакт з багатьма людьми, наявність атопічного дерматиту, який знижує захисні функції епідермісу, а також можливий вплив косметичних процедур за кілька тижнів до захворювання. Цей факт ще раз доводить необхідність санітарного контролю та навчання дотримання правил асептики та антисептики працівників закладів, пов’язаних з наданням косметологічних та загальних гігієнічних послуг: перукарень, косметологічних, масажних кабінетів, кабінетів татуажу тощо. Крім того, дерматологам при консультуванні пацієнтів з атопічним дерматитом слід наголошувати на виконанні засобів профілактики мікробних, вірусних та грибкових інфекцій: щеплення, особливо проти вітряної віспи.
Другим аспектом є незвичайна етіологія мікробної інфекції шкіри в цієї пацієнтки, яка жодного разу не перебувала на лікуванні в лікарні та за останні кілька років не приймала антибіотики. Початок захворювання за клінічними ознаками вкрай нагадував локальну форму стрептодермії або стафілодермії, що мало ризик розвитку системної інфекції. Водночас ефективне лікування було досягнуто завдяки великим дозам напівсинтетичного антибіотика групи цефалоспоринів 1 покоління – цефазоліну. Однак через короткий проміжок часу стався рецидив захворювання.
Ураховуючи клініко-лабораторні дані та анамнез, було сформовано три «клінічні парадигми»: 1) резистентний збудник, 2) мікробна суперінфекція шкіри, 3) виникнення імунопатологічного стану, схожого на постстрептококовий пустульоз (наявність поодиноких елементів висипу на долонях) на фоні атопії шкіри [15]. Тому, не чекаючи результатів мікробіологічного дослідження, було ухвалено рішення застосувати антибіотик резерву з класу оксазолідинонів – лінезолід, з огляду на його фармакологічний спектр впливу на S. aureus, S. pyogenes та E. faecalis. Зважаючи на атопічний дерматит і можливий імунопатологічний компонент у виникненні рецидиву, був призначений глюкокортикостероїдний (ГКС) препарат преднізолон системно, внутрішньо, ультракоротким курсом на 5 днів. Надзвичайно важливим у виборі протимікробної терапії та доцільності застосування ГКС є валідність мікробіологічних досліджень. За результатами повторних мікробіологічних тестів у пацієнтки була виявлена суперінфекція: перша (до призначення антибіотиків) – мікст-інфекція S. agalactiae та S. aureus, друга (після першого епізоду) – E. faecalis під час рецидиву. Важливо зазначити, що пацієнтка весь час дотримувалась усіх гігієнічних норм. Доведено, що небульозне імпетиго може бути спричинене β-гемолітичними стрептококами групи А (S. pyogenes) або S. aureus, або їх комбінацією [16, 17, 18]. Щодо S. agalactiae, то описані рідкісні випадки бульозного імпетиго, спричиненого стрептококами групи В в новонароджених [7]. Ці нетипові неонатальні ураження шкіри пов'язані з колонізацією родових шляхів матері стрептококами групи В. Також E. faecalis є нетиповим збудником інфекцій шкіри цієї локалізації та глибини ураження тканин для дорослих [15, 16]. Згідно з дослідженнями, ентерококові інфекції ран і м’яких тканин більш поширені в травматологічних установах і можуть бути пов'язані з інфекціями після операцій, що стосуються шлунково-кишкового тракту або жіночих статевих шляхів [5, 8]. Тому виявлення S. agalactiae та E. faecalis є нетиповим та потребує вивчення.
Третім аспектом є комунікативний. Зрозуміло, що пацієнти хочуть швидко одужати без значних витрат, і це може бути фактором втрати довіри до лікарів, чому також сприяє вільний доступ до медичної інформації через мережу «Інтернет», яка часто містить непрофесійні або популістські (комерційні) матеріали. Ця проблема стає актуальною, коли пацієнти не мають чіткого розуміння причин захворювання, які неможливо виявити без певних лабораторних та інструментальних досліджень.
ВИСНОВКИ
1. Атопічний дерматит та професійні ризики щодо контактних інфекцій є суттєвими факторами розвитку тяжких мікробних уражень шкіри.
2. Наявність у лікаря результатів валідних лабораторних досліджень є об’єктивним аргументом для ефективного лікування.
3. Відсутність вищеозначеного може спонукати до самолікування, збільшити ризик ускладнень, поширення інфекцій і зменшити довіру до системи громадського здоров’я.
Практичні рекомендації
1. Включення мікробних інфекцій шкіри та підшкірної клітковини до обов’язкової реєстрації у звіт «Інфекційної захворюваності населення України» сприятиме боротьбі з поширенням стійкості до протимікробних препаратів, як елемента системи контролю інфекцій.
2. Атопія шкіри та «ризиковані» професії, що пов’язані з ризиком контактних інфекцій, є суттєвими факторами для виникнення тяжких мікробних інфекцій шкіри.
3. Валідний мікробіологічний контроль мікробних інфекцій шкіри є визначальним інструментом для вибору терапії та об’єктивної оцінки її ефективності.
4. Наявність у лікаря валідних лабораторних досліджень є об’єктивним аргументом для комунікації з пацієнтом, що дозволить покращити результати лікування, зменшити витрати й ризик позовів/скарг з приводу некваліфікованого лікування.
5. Забезпечення мікробіологічних лабораторій необхідним обладнанням та витратними матеріалами; навчання персоналу та участь у зовнішньому контролі якості; налагодження комунікації між мікробіологічною лабораторією та клінічними відділеннями, особливо на преаналітичному етапі; а також створення кумулятивних антибіотикограм мають вирішальне значення для отримання якісних та своєчасних результатів мікробіологічних досліджень.
Внески авторів:
Маврутенков В.В. – методологія, написання – початковий проєкт, написання – рецензування та редагування;
Литвин К.Ю. – концептуалізація, написання – рецензування та редагування;
Якуніна О.М. – написання – початковий проєкт;
Будаєва І.В. – візуалізація, курація даних;
Ювко О.В. – дослідження, ресурси.
Фінансування. Дослідження не має зовнішніх джерел фінансування.
Конфлікт інтересів. Автори заявляють про відсутність конфлікту інтересів.
REFERENCES
1. Stoneham S, Peters J, Price J. Staphylococcal and streptococcal infections. In: Stoneham S, Peters J, Price J, editors. Habif’s Clinical Dermatology: A Colour Guide in Diagnosis and Therapy. 7‑th ed. Elsevier; 2021. p. 731-8. doi: https://doi.org/10.1016/j.mpmed.2021.09.001
2. [Ministry of Health of Ukraine. Annual report on the health status of the population of Ukraine and the epidemic situation for 2022]. Kyiv; 2023. Ukrainian.
3. Lancefield RC. A serological differentiation of human and other groups of hemolytic streptococci. J Exp Med. 1933;57(4):571-95. doi: https://doi.org/10.1084/jem.57.4.571
4. Lancefield RC, Hare R. The serological differentiation of pathogenic and non‑pathogenic strains of hemolytic streptococci from parturient women. J Exp Med. 1935;61(3):335-49. doi: https://doi.org/10.1084/jem.61.3.335
5. Fry RM. Fatal infections by haemolytic Streptococcus group B. Lancet. 1938;1:199-201. doi: https://doi.org/10.1016/S0140-6736(00)93202-1
6. Braunstein H, Tucker EB, Gibson BC. Identification and significance of Streptococcus agalactiae (Lancefield group B). Am J Clin Pathol. 1969;51(2):207-13. doi: https://doi.org/10.1093/ajcp/51.2.207
7. Eickhoff TC, Klein JO, Daly AK, Ingall D, Finland M. Neonatal sepsis and other infections due to group B beta‑hemolytic streptococci. N Engl J Med. 1964;271:1221-8. doi: https://doi.org/10.1056/NEJM196412102712401
8. Lazarus JM, Sellers DP, Marine WM. Meningitis due to the group B beta‑hemolytic Streptococcus. N Engl J Med. 1965;272:146-7. doi: https://doi.org/10.1056/NEJM196501212720308
9. Mannik M, Baringer JR, Stokes J III. Infections due to group B beta‑hemolytic streptococci. Report of three cases and review of the literature. N Engl J Med. 1962;266:910-3. doi: https://doi.org/10.1056/NEJM196205032661803
10. Hall LM, Gorges HJ, van Driel M, Magin P, Francis N, Heal CF. International comparison of guidelines for management of impetigo: a systematic review. Fam Pract. 2022 Feb;39(1):150-8. doi: https://doi.org/10.1093/fampra/cmab066
11. [On approval of reporting forms for infectious and parasitic diseases, vaccinations against certain infectious diseases and instructions for filling them out. Order of the Ministry of Health of Ukraine dated 2009 Jun 02 No. 378]. [Internet]. 2009 [cited 2026 Jan 03]. Ukrainian. Available from: https://zakon.rada.gov.ua/laws/show/z0525-09#Text
12. [State strategy for combating antimicrobial resistance for the period until 2030. Order of the Cabinet of Ministers of Ukraine dated 2024 Dec 13 No. 1265-r]. [Internet]. 2024 [cited 2026 Jan 03]. Ukrainian. Available from: https://zakon.rada.gov.ua/laws/show/1265-2024-%D1%80#n19
13. European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 14.0.2024 [Internet]. [cited 2026 Jan 03]. Available from: https://www.megumed.de/wp-content/uploads/2024/02/v_14.0_Breakpoint_Tables.pdf
14. Global antibiotic resistance surveillance report 2025. World Health Organization [Internet]. 2025 Oct 13 [cited 2026 Jan 03]. Available from: https://www.who.int/publications/i/item/9789240116337
15. Wang V, van Rensburg PJJ, Boguniewicz J, Ong PY. Infectious complications of atopic dermatitis. An update. Ann Allergy Asthma Immunol. 2025;135(5):487-97. doi: https://doi.org/10.1016/j.anai.2025.05.021
16. Nardi NM, Schaefer TJ. Impetigo [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jul 31 [cited 2026 Jan 03]. Available from: www.ncbi.nlm.nih.gov
17. Impetigo: antimicrobial prescribing. NICE [Internet]. 2020 Feb 26 [cited 2026 Jan 05]. Available from: https://www.nice.org.uk/guidance/ng153
18. CLSI. Analysis and Presentation of Cumulative Antimicrobial Susceptibility Test Data [Internet]. 5 th ed. 2022 [cited 2026 Jan 03]. Available from: https://nms.go.ug/wp-content/uploads/2025/12/CLSI_M39Ed5E-standard_Analysis-and-Presentation-of-Cumulative-AST-data1.pdf
Key words: premature ejaculation, erectile dysfunction, combat injury, venlafaxine, dapoxetine
Ключові слова: передчасна еякуляція, еректильна дисфункція, бойова травма, венлафаксин, дапоксетин
Abstract
Premature ejaculation is one of the most common male sexual disorders and occurs with a frequency of 21-33% in various populations. The etiology of premature ejaculation is practically not established, although it is a fairly common type of sexual dysfunction. According to various data, sexual dysfunction occurs in 49.1-80% veterans of combat-actions with post-traumatic stress disorder. Premature ejaculation can interfere with sexual satisfaction, leading to a reduced quality of life for men and their partners. It has been shown that serotonin reuptake inhibitors may be the drugs of choice in the treatment of premature ejaculation. The aim of the work was to improve methods of treating sexual dysfunction accompanied by premature ejaculation in men injured as a result of hostilities. 56 men injured as a result of hostilities with sexual dysfunction and complaints of premature ejaculation were examined. Patients were divided into two groups depending on the selected selective serotonin reuptake inhibitors – venlafaxine (n=24) and dapoxetine (n=32) which they had been receiving for at least 1.5 months. After treatment with both serotonin reuptake inhibitors, reactive and personality anxiety symptoms objectively decreased, as assessed by the Spielberger-Hanan scale. Treatment with venlafaxine and paroxetine resulted in a significant reduction in depressive symptoms in men with premature ejaculation. They turned out to be relatively balanced drugs with satisfactory efficacy and relatively few side effects. However, venlafaxine, compared to dapoxetine, produced 2.1 times fewer side effects and 20% fewer relapses. The average duration of sexual intercourse after treatment with both drugs positively correlated with the overall improvement of sexual function according to the IIEF-5 scale (r=0.70; p<0.05 for the first group; r=0.85; p<0.05 for the second group). An inverse correlation was found between the average duration of sexual intercourse and the score of anxiety symptoms on the Spielberger-Hanin scale in patients of both study groups (r= -0.69; p<0.05 for the first group; r= -0.68; p<0.05 for the second group). Although dapoxetine is considered the “gold standard” among serotonin reuptake inhibitors for the treatment of premature ejaculation, the drug is used by the patient only on demand, and other drugs in this group, which are antidepressants, may have a more positive effect on patients with depression and concomitant premature ejaculation, who have suffered as a result of hostilities. The study demonstrates that the neurotransmitter serotonin plays a key role in the modulation of ejaculation, as the use of its reuptake inhibitors, in particular dapoxetine and venlafaxine, contributes to an increase in intravaginal latency time to ejaculation.
Реферат
Порівняльний ефект дапоксетину та венлафаксину в лікуванні передчасної еякуляції чоловіків, постраждалих унаслідок бойових дій. Воробець М.З., Воробець Д.З., Фафула Р.В., Мельник О.В., Личковська Н.Е., Воробець З.Д., Чемерис О.М. Передчасна еякуляція є одним з найбільш поширених чоловічих статевих розладів і зустрічається з частотою 21-33% у різних популяціях. Етіологія передчасної еякуляції практично не встановлена, хоча вона є досить поширеним видом сексуальної дисфункції. За різними даними, у ветеранів бойових дій із посттравматичним стресовим розладом сексуальна дисфункція зустрічається в 49,1-80%. Передчасна еякуляція може перешкоджати сексуальному задоволенню, що призводить до зниження якості життя чоловіків та їхніх партнерок. Показано, що саме інгібітори зворотного захоплення серотоніну можуть бути препаратами вибору при лікуванні передчасної еякуляції. Метою роботи було удосконалення методів лікування статевої дисфункції, що супроводжується передчасною еякуляцією, у чоловіків, постраждалих унаслідок бойових дій. Обстежено 56 чоловіків, постраждалих унаслідок бойових дій, із сексуальною дисфункцією та скаргами на передчасну еякуляцію. Пацієнтів розподілено на дві групи залежно від вибраного препарату селективного інгібітора зворотного захоплення серотоніну, який вони отримували щонайменше 1,5 місяці: венлафаксин (n=24) та дапоксетин (n=32). Після лікування обома інгібіторами зворотного захоплення серотоніну в чоловіків об’єктивно зменшилась реактивна та особистісна тривожна симптоматика, яка оцінювалась за шкалою Спілбергера-Ханана. Лікування венлафаксином і дапоксетином привело до вірогідного зменшення депресивної симптоматики в чоловіків з передчасною еякуляцією. Вони виявились відносно збалансованими препаратами із задовільною ефективністю і відносно невеликою кількістю побічних ефектів. Однак дапоксетин, порівняно з венлафаксином, давав у 2,1 раза меншу кількість побічних ефектів і на 20% менше рецидивів. Середня тривалість статевого акту після лікування обома препаратами позитивно корелює із загальним покращенням сексуальної функції за шкалою МІЕФ-5 (r=0,70; p<0,05 для першої групи; r=0,85; p<0,05 для другої групи). Обернена кореляція виявлена між середньою тривалістю статевого акту та балом тривожної симптоматики за шкалою Спілбергера-Ханіна в пацієнтів обох досліджуваних груп (r= -0,69; p<0,05 для першої групи; r= -0,68; p<0,05 для другої групи). Хоча «золотим стандартом» серед препаратів інгібіторів зворотного захоплення серотоніну для лікування передчасної еякуляції вважається дапоксетин, препарат вживається пацієнтом лише за вимогою, а інші препарати цієї групи, які відносяться до антидепресантів, можуть мати також позитивний вплив саме на пацієнтів з депресією та супутньою передчасною еякуляцією, постраждалих унаслідок бойових дій. Дослідження демонструє, що нейротрансмітер серотонін відіграє ключову роль у модуляції еякуляції, оскільки застосування інгібіторів його зворотного захоплення, зокрема дапоксетину та венлафаксину, сприяє збільшенню інтравагінального латентного часу до настання еякуляції.
Men with sexual dysfunction often complain of premature ejaculation (PE) which is considered as the inability to control ejaculation for “enough” time to have sexual intercourse [1, 2, 3]. Premature ejaculation is one of the most common male sexual disorders and occurs with a frequency of 21-33% in different populations [2, 4]. According to other data, 25-40% of men in the USA suffer from PE [5, 6]. The etiology of PE is practically not established, although it is a fairly common type of sexual dysfunction.
According to various data, sexual dysfunction occurs in 49.1-80% of combat veterans with posttraumatic stress disorder [7, 8, 9]. PE can interfere with sexual satisfaction, leading to a decrease in the life quality of patients and their partners [1, 3]. The mechanism of PE development is not fully understood, especially considering the role of physiological mechanisms of action of selective serotonin reuptake inhibitors (SSRI). SSRI were introduced into the treatment of PE after psychopharmacological studies on the pathways of ejaculation control [1, 2, 10].
Psychological factors often influence the occurrence of PE. Men sometimes underestimate the connection between sexual ability and emotional well-being. PE can be caused by temporary depression or stress. However, it is often necessary to use drugs that slow down the rate of arousal. Serotonin reuptake inhibitors have been indicated as the drugs of choice in the treatment of PE [1-5]. Along with behavioral therapy, this paper presents our comparative experience with the use of medications in this group. Clinical and laboratory studies of sexual dysfunction in recent years have led to the development of new treatment protocols, including psychosexual and local negative pressure therapy methods, and new pharmacological drugs [1, 3, 11].
Dapoxetine hydrochloride, a short-acting selective serotonin reuptake inhibitor, is currently the only oral drug recommended on label [1, 2, 12-15]. The mechanism of action of dapoxetine in PE is likely related to the inhibition of serotonin reuptake by neurons and the subsequent enhancement of the effects of neurotransmitters on pre- and postsynaptic receptors. Analysis of clinical studies has confirmed that this on-demand treatment improves the quality of life of the patient and his sexual partner [1, 10]. However, the drug is taken only on demand, and is often viewed by patients as a "one-off", that is, one that does not treat the causes of the disease and cannot potentially cure it. Dapoxetine causes the most significant delay in ejaculation among SSRI, which are antidepressants, and is therefore widely used for the treatment of PE [18]. Although dapoxetine was originally developed as an antidepressant, it has been largely unused for the treatment of depression and is now only used for the treatment of PE [19].
Patients who have suffered as a result of hostilities and who are consulted by a psychiatrist often exhibit depressive symptoms and are prescribed antidepressants. Some of the antidepressants that belong to the SSRI group also have a positive effect on prolonging the intravaginal latency time before ejaculation. Therefore, one drug can be successfully used by a psychiatrist and a sexologist to treat depression and concomitant PE.
Venlafaxine is an antidepressant from the group of selective serotonin and norepinephrine reuptake inhibitors, also used to treat PE [16, 17].
All forms of ejaculatory disorders require a comprehensive psychological examination of patients using objective questionnaire methods, and the development of modern comprehensive therapy methods, which will ensure better cure rates. In this study, we sought to evaluate the treatment effect of dapoxetine compared with venlafaxine on combat veterans with combat injuries accompanied by depressive and sexual disorders, in particular PE in real-world practice and to investigate factors influencing treatment effectiveness. The aim of the work was to improve methods of treating sexual dysfunction accompanied by premature ejaculation in men injured as a result of hostilities.
MATERIALS AND METHODS OF RESEARCH
The research was conducted at the Urology Clinic of the Danylo Halytsky Lviv National Medical University and the Military Medical Clinical Hospital in the Western Region. The work was based on the results of the examination and treatment of 56 men injured as a result of hostilities, with sexual dysfunction and complaints of premature ejaculation. The age of the patients ranged from 20 to 53 years, with a mean age of 39±5.2 years. All patients completed and signed an informed consent to participate in the studies. The research was carried out in compliance with all principles of medical ethics and protection of patients' rights, human dignity and moral and ethical norms, in accordance with the principles of the Helsinki Declaration of Human Rights, the Council of Europe Convention on Human Rights and Biomedicine, the Laws of Ukraine that determine this; permission of the Bioethics Commission of the Danylo Halytskyi Lviv National Medical University (protocol No. 7 of 26.06.2023).
All patients underwent a questionnaire based on the IIEF-5 scale (International Index of Erectile Function-5) [20, 21], a sexological and urological history was collected, a rectal examination of the prostate was performed, microscopy of the prostate secretion for the presence of an inflammatory process, prostate ultrasonography, and, if indicated, bacteriological culture of the prostate secretion, and, if necessary, PCR diagnostics of the relevant pathogens in the prostate secretion.
To determine health-related quality of life (QOL), a survey was conducted using the SF-36 questionnaire [22, 23, 24]. According to the etiological principle, neurogenic genesis of PE was found among all examined patients. Patients were routinely consulted by a psychologist, sexologist, and psychiatrist. The main complaint of patients was PE, which persisted with regular sexual activity 2.4±0.3 times/week. Patients were assigned to this group in the absence of complaints, anamnesis, clinical, instrumental and laboratory data of the presence of prostatitis (colliculoprostatitis) or chronic pelvic pain syndrome. All patients of this group had a pathological fixation on sexual dysfunction, which was accompanied by an affective component in the form of anxiety, fear of failure, bad mood, and intense doubts, and led to the dominance of the sympathetic nervous system during sexual intimacy [1, 11, 12]. The average duration of the disease was 9.0±0.8 months.
Thus, the study group included only men with neurogenic (psychogenic) PE, which in all 56 (100%) occurred after a combat injury (multiple shrapnel and bullet wounds). Given the complexity of the pathogenesis of PE, the treatment of all patients was comprehensive and consisted of two parts: basic therapy and selective serotonin reuptake inhibitors (SSRI).
Patients were divided into two groups depending on the chosen SSRI drug, which they received for at least 1.5 months: group 1 (n=24) received venlafaxine, 37.5-150 mg/day; group 2 (n=32) received dapoxetine, 30-60 mg/day.
To objectively reflect anxiety and depression, patient questionnaires were used using the Spielberger-Hanin and Hamilton scales, respectively [24]. During the treatment process, criteria for determining erectile dysfunction were taken into account.
To assess the effectiveness of treatment methods for patients with ejaculatory disorders, the obtained digital data were processed by statistics methods using parametric and nonparametric multiple comparison methods. The results are presented as arithmetic means with standard errors (M±m) and medians with 25% and 75% percentiles (Me [Q25; Q75]). The reliability of differences between independent quantitative variables with a normal distribution was assessed using the Student (t) and Mann-Whitney U-test. Differences between the study groups were considered statistically significant at p<0.05. The correlation was calculated by determining the Spearman's rank correlation coefficient [24, 25]. Mathematical processing of the results was carried out using the application package of licensed statistical program Microsoft Excel (Microsoft Office Standard 00218-04988-70316-AA857).
RESULTS AND DISCUSSION
Treatment of men with premature ejaculation was included in the study. Men with sexual dysfunction usually have impaired sexual motivation: instead of focusing on the process of lovemaking, patients are focused on the fact that sexual intercourse may end prematurely. They experience tension and anxiety instead of positive emotions. Given this, an important component of the success of the treatment was the patient's ability to learn to clearly navigate their feelings and apply a functional training option in a timely manner in order to prevent the ejaculatory reflex from being triggered. The effectiveness of PE treatment in patients of both SSRI groups, expressed in an increase in the duration of sexual intercourse (IELT), the presence and severity of side effects, as well as PE recurrences, is shown in Table.
Thus, before the start of treatment, the average duration of sexual intercourse in group 1 was 1.0±0.4 minutes. In all 24 patients of group 1, therapy was initiated with a dose of venlafaxine 37.5 mg in the evening for 2-4 days, followed by a transition to 37.5 mg twice a day to prevent nausea, which was observed with varying severity up to the first 5 days of administration in 16/24 (66.7%), and then completely disappeared in 22/24 men. One patient refused to continue using the drug.
Most patients experienced moderate general weakness and fatigue a few days after the start of treatment, which was difficult to objectify due to the significant polymorphism of subjective sensations. Moderate decreased libido and mild erectile dysfunction were noted by 8/24 (33%) men. The positive side is the rapid increase of the intravaginal latency time to ejaculation (IELT) within 5-7 days of taking venlafaxine. The negative side is that the drug is the most expensive in the SSRI segment. By 1.5 months of treatment, 18/22 (81.8%) men reported significant subjective improvement with an average increase in IELT of 3.0±0.45 min; two others (18.2%), not satisfied with the duration of IELT, switched to a dose of 150 mg twice a day and after 2 weeks reported an IELT of 5.0±0.12 min. Therefore, the overall effectiveness of venlafaxine in patients of our clinic was almost 100% and depended only on the dose received. However, the recurrence of PE after drug withdrawal was 18/24 (75%). Some patients indicated a positive effect (increased IELT) from taking venlafaxine "on demand", a few hours before the expected sexual intercourse, but these data cannot be reliably objectified. 10/24 (41.7%) patients agreed to continue taking venlafaxine after 1.5 months.
In group 2, before the start of treatment, the average duration of sexual intercourse was 1.0±0.4 min. In all 32 patients in group 2, therapy was initiated with a dose of dapoxetine 20 mg/day in the afternoon, preferably after work, for 45 days, which was well tolerated. Among the side effects during the first few days of taking the drug, the most common were nausea, dizziness, headache, and diarrhea for 1-3 days in 10/32 (31.25%) men, which usually resolved quickly. Moderate decreased libido and mild erectile dysfunction were noted by 8/32 (25%) men. After 1.5 months, 26/32 (81.25%) men reported significant subjective improvement with an average increase in IELT to 3.0±0.34 min; 4/32 (12.5%) reported slight improvement with IELT to 2±0.12 min, and 2/32 (6.25%) reported no increase in the duration of sexual intercourse with IELT of 1.0±0.03 min.
Groups > group 1 (n=24) venlafaxine 75 mg/day group 2 (n=32) dapoxetine 30 mg/day Treatment > until after until after Average duration of sexual intercourse (intravaginal latency time to ejaculation (IELT)), min, (M±m, Me[Q25; Q75]) 1.0±0.4 1.2 [0.8; 1.3] 3.0±0.45* 3.4 [3.2; 3.7] (81.8% - significant improvement) 5.0±0.12* 4.9 [4.8; 5.1] (18.2% - significant improvement after increasing the dose 1.0±0.4 1.2 [0.8; 1.3] 3.0±0.34* 3.3 [3.0; 3.8] (81.25% - significant improvement) 2.0±0.12* 2.1 [2.0; 2.3] (12.5% - slight improvement after increasing the dose 1.0±0.03 1.0 [1.0; 1.1] (6.25% - no improvement) total efficiency 93.75% Adverse effects, % 66.70 31.25 Decreased libido and mild ED, % 33 25 Overall improvement in sexual performance on the IIEF scale across all domains, number of points 5.4 6.2 Depressive symptoms according to the Hamilton scale, points 6 5 6 5 Anxiety symptoms according to the Spielberger-Hanin scale, reactive/personal, scores 34/31 15/9* 34/31 15/9* Patients who wish to continue treatment for more than 1.5 months, % 41.70 43.75 PE recurrence after drug withdrawal, % 75.0 62.5 Notes: * the difference with the indicator before treatment is significant, p<0.05.
to 150 mg/d) total efficiency 100%
to 40 mg/d)
The latter agreed to increase the dose of paroxetine to 40 mg/day over the next month. Only five (31.25%) men who experienced significant improvement and two (12.5%) who noted moderate improvement – a total of 43.75% patients decided to continue treatment with paroxetine for more than 1.5 months, explaining this by the same reluctance to commit to systematic pill use. Different respondents noted an increase in IELT from 1 week to 1 month of dapoxetine use. Episodic use of this drug did not lead to a significant prolongation of the duration of coitus. The recurrence of PE from the first days of drug withdrawal to six months was 20/32 (62.5%). Overall, patients were fairly satisfied with dapoxetine, given its average effectiveness, average cost, and minor side effects compared to other SSRI.
Overall, the results obtained indicate the effectiveness of treating PE with serotonin reuptake inhibitors (Fig. 1).
The vast majority of patients ultimately found medication treatment psychologically difficult, as it only helps with systematic use and does not provide a lasting effect after discontinuation. The fact that the total IIEF-5 score increased by 5.4 and 6.2 after treatment with venlafaxine and paroxetine, respectively, turned out to be interesting (Fig. 2).
This once again indicates the close relationship between all components of sexual function, when, with the improvement of the ability to control ejaculation, the scores of the IIEF domains, which reflect the improvement of sexual function in general, simultaneously improve. Also, after treatment with SSRI, anxiety in patients with PE significantly decreased, as calculated using the Hamilton scale, which in turn confirms the close relationship between PE, psychogenic symptoms at the clinical level, and the state of serotonergic transmission of excitation at the physiological level. The most pronounced trend towards reduced anxiety is observed after the use of dapoxetine.
After treatment with both SSRI, reactive and personal anxiety symptoms objectively decreased, as assessed by the Spielberger-Hanan scale. However, it is difficult to carry out an absolutely correct comparative analysis of the effectiveness of both drugs, since the groups included men with very different self-assessments of anxiety. Some patients denied it, others noted pronounced anxiety, which was most likely associated with sexual disorders.
Treatment with venlafaxine and dapoxetine resulted in a significant reduction in depressive symptoms in men with PE, but this was more dependent on the even distribution of patient groups, many of whom, despite PE, denied any depression.
Men with PE always hope for a "complete cure," which, unfortunately, is not often possible at current stage of development of sexology, psychiatry, and pharmacology. This necessitates the search for new, more effective treatments.
The correlation coefficient was used to determine the relationships between overall improvement in sexual function on the IIEF scale across all domains and average duration of sexual intercourse. The average duration of sexual intercourse after treatment with both drugs is positively correlated with the overall improvement in sexual function according to the
IIEF-5 scale (r=0.70; p<0.05 for the first group; r=0.85; p<0.05 for the second group) (Fig. 3). An inverse correlation was found between the average duration of sexual intercourse and the score of anxiety symptoms on the Spielberger-Hanin scale in patients of both study groups (r= -0.69; p<0.05 for the first group; r= -0.68; p<0.05 for the second group) (Fig. 4).
Thus, the study results indicate an association between combat-related trauma in military personnel and the development of neurogenic (psychogenic) premature ejaculation. According to sexual, urological, and psychoneurological history data, premature ejaculation was not reported prior to participation in combat activities. These results are, to some extent, consistent with those obtained in a study of the impact of stress factors (without injuries) on the civilian population in Israel, resulting from combat operations on sexual dysfunction and, in particular, on PE, which was observed in 56.5% of men [26]. Probably, in our case, the causal pathophysiological relationship between premature ejaculation and the consequences of combat operations is mediated by psychological (stress, anxiety, hyperarousal, development of post-traumatic stress disorder), neurophysiological and hormonal mechanisms, as well as, importantly, by medical treatment of wounds. At the same time, it is obvious that additional research is needed to accurately clarify the mechanisms of premature ejaculation development: psychological, neurophysiological, hormonal, biochemical, and possibly immunological, and others.
The study demonstrates that the neurotransmitter serotonin plays a key role in the modulation of ejaculation, as the use of its reuptake inhibitors promotes the increase in intravaginal latency time to ejaculation.
Among SSRI, venlafaxine and dapoxetine were almost equally effective when used together. However, paroxetine caused significantly fewer side effects (31.25%) compared to venlafaxine (66.7%). Also, dapoxetine resulted in lower recurrence of PE (62.5%) compared to venlafaxine (75.0%). Although, in both cases, after the end of the course of treatment, most patients experienced a recurrence of PE.
The use of selective serotonin reuptake inhibitors leads to an increase in the time to ejaculation [13, 23]. In recent years, the most commonly used drugs for the treatment of PE are Longride, Priligy, and Daporin, but the active ingredient in all of them is Dapoxetine. Before the introduction of dapoxetine, many SSRI were used, but their effectiveness was much lower, since permanent use did not guarantee a stable effect, and long-term use caused the development of side effects [27]. Dapoxetine is the first drug specifically designed for the treatment of PE and has high selectivity [1, 14, 15], although it was primarily developed as an antidepressant [19]. Unlike other SSRI, dapoxetine has no significant clinical electrocardiographic effects, is safe and well tolerated at doses of 30-60 mg/day, although it is prescribed only on demand and cannot potentially cure the psychogenic cause of PE, unlike long-term SSRIs, especially in patients with concomitant depressive symptoms [1].
Some clinical studies previous years indicate of significant efficacy of paroxetine [23, 28, 29]. It has been shown that 77.8% of patients with lifelong PE decided to continue daily dapoxetine use after 3 months of successful use, 30.8% refused due to unsatisfactory efficacy [3, 13].
A multicenter study of married couples over 6 months in 5 countries used stopwatches to measure intravaginal latency time to ejaculation, the period from intromission to ejaculation [3, 30]. It was found that the IELT ranged from 30 seconds to 44 minutes, with an average of 5.4 minutes; also depending on the country – from 3.7 minutes to 7.6 minutes. In more than 14% of men, the average IELT was lower than 200 s, in 26% it was longer than 600 s.
Clomipramine may help in severe cases of PE associated with serious nervous system disorders; the drug has also been reported to improve erection quality in some patients [23]. Intramuscular injections of magnesium sulfate solution have been reported to be successful, as PE is believed to be associated with magnesium deficiency [13, 14]. The use of the LORETA (low-resolution electromagnetic tomography of the brain) functional imaging method in patients revealed that the SSRI sertraline promotes an increase in electrical activity mainly in the frontal, limbic and temporal lobes of the left hemisphere of the brain, which may be associated with the therapeutic effect of SSRI in PE [31, 32, 34].
Some studies have shown that PE in men from different countries of the world occurs even more often than erectile dysfunction, accounting for 20-30% of the population and practically not changing with age [11]. PE not only affects the sexual health of the man, but also negatively affects the psychosocial health of the partner [23, 35].
Due to the polyetiological nature of PE, the involvement in the development of PE of not only anatomical and physiological links of pathogenesis, but also socio-psychological aspects, as well as taking into account the paired nature of sexual function, certain difficulties arise in finding effective comprehensive methods of treating PE at the current stage of development of medicine.
Thus, the study provides new data on the pharmacotherapeutic features of treating premature ejaculation in male combatants with dapoxetine and venlafaxine, contributing to a better understanding of the efficacy and tolerability of selective serotonin reuptake inhibitors in the context of increased anxiety.
CONCLUSIONS
1. Among the serotonin reuptake inhibitors, venlafaxine (100%) was more effective in treating men with premature ejaculation who were injured as a result of combat operations than dapoxetine (93.75%), but no significant difference was found between them (p>0.05).
2. The use of dapoxetine causes fewer side effects (31.25%) in the treatment of premature ejaculation than venlafaxine (66.7%). Also, dapoxetine causes fewer relapses (62.5%), compared to venlafaxine (75%).
3. The average duration of sexual intercourse after treatment of premature ejaculation with dapoxetine and venlafaxine is positively correlated with the overall improvement of sexual function according to the MIEF-5 scale (r=0.70; p<0.05 for the first group; r=0.85; p<0.05 for the second group). An inverse correlation was found between the average duration of sexual intercourse and the score of anxiety symptoms according to the Spielberger-Khanin scale in men of both study groups (r= -0.69; p<0.05 for the first group; r= -0.68; p<0.05 for the second group) injured as a result of hostilities.
4. Due to the absence of a “gold standard” for the treatment of premature ejaculation, dapoxetine and venlafaxine, among selective serotonin reuptake inhibitors, demonstrated comparable efficacy in the treatment of premature ejaculation (with a significant increase in intravaginal ejaculatory latency time) in male combatants, without extrapolation of the results to other therapeutic approaches.
Contributions:
Vorobets M.Z. – treatment, methodology, writing – initial draft;
Vorobets D.Z. – conceptualization, treatment, data curation, writing – review & editing, resources;
Fafula R.V. – formal analysis, verification;
Melnyk O.V. – visualization, formal analysis;
Lychkovska N.E. – visualization;
Vorobets Z.D. – supervision, project administration;
Chemerys O.M. – date curation, project administration.
Funding.The research was conducted with the financial support of a grant from the National Research Foundation of Ukraine “Improving the diagnostics and treatment of sexual and reproductive disorders (disorders) in men affected by hostilities” (registration No. 2022.01/0151).
Conflict of interest. The authors declare no conflict of interest.
REFERENCES
1. Liu G, Yin Y, Zhang L, He D, Yang L. Efficacy of Dapoxetine in the Treatment of Patients With Lifelong Premature Ejaculation as an Alternative to Sertraline Therapy. Sexual Medicine. 2022;10(1):100473. doi: https://doi.org/10.1016/j.esxm.2021.100473
2. Peng J, Fang D, Li H, et al. Efficacy of dapoxetine treatment in Chinese patients with premature ejaculation and possible factors affecting efficacy in the real-world practice. BMC Urol. 2020;20:11. doi: https://doi.org/10.1186/s12894-0200580-3
3. Salonia A, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, et al. European Association of Urology Guidelines on Sexual and Reproductive Health-2021 Update: Male Sexual Dysfunction. Eur Urol. 2021 Sep;80(3):333-57. doi: https://doi.org/10.1016/j.eururo.2021.06.007
4. Gillman N, Gillman M. Premature Ejaculation: Aetiology and Treatment Strategies. Medical Sciences. 2019;7(11):102. doi: https://doi.org/10.3390/medsci7110102
5. Dahm P, Holten M, Ratanawong JP, Lauwagie A, Gonzalez‐Padilla DA. Analysis of European Association of Urology Guidelines 2023 and its adherence to GRADE methodology. Clin Pub Health Guidelines. 2024;1:e70004. doi: https://doi.org/10.1002/gin2.70004
6. Crowdis M, Leslie SW, Nazir S. Premature Ejaculation. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 [cited 2025 Mar 23]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK546701/
7. Bird ER, Piccirillo M, Garcia N, Blais R, Campbell S. Relationship Between Posttraumatic Stress Disorder and Sexual Difficulties: A Systematic Review of Veterans and Military Personnel. J Sex Med. 2021 Aug;18(8):1398-426. doi: https://doi.org/10.1016/j.jsxm.2021.05.011
8. Breyer BN, Fang SC, Seal KH, et al. Sexual Health in Male and Female Iraq and Afghanistan U.S. War Veterans With and Without PTSD: Findings From the VALOR Cohort. Journal of Traumatic Stress. 2016 Jun;29(3):229-36. doi: https://doi.org/10.1002/jts.22097
9. Castillo OJr, Chen IK, Amini E, Yafi FA, Barham DW. Male Sexual Health Related Complications Among Combat Veterans. Sex Med Rev. 2022 Oct;10(4):691-7. doi: https://doi.org/10.1016/j.sxmr.2022.06.002
10. El-Hady AK, El-Gamil DS, Abdel-Halim M, Abadi AH. Advancements in Phosphodiesterase 5 Inhibitors: Unveiling Present and Future Perspectives. Pharmaceuticals (Basel). 2023 Sep 6;16(9):1266. doi: https://doi.org/10.3390/ph16091266
11. DeLay KJ, Haney N, Hellstrom WJ. Modifying Risk Factors in the Management of Erectile Dysfunction: A Review. World J Mens Health. 2016 Aug;34(2):89-100. doi: https://doi.org/10.5534/wjmh.2016.34.2.89
12. Zhong C, Li C, Geng Q, Han Q, Gao Q, Zhang J, et al. Reasons and treatment strategy for discontinuation of dapoxetine treatment in premature ejaculation patients in China: A retrospective observational study. Andrologia. 2022 Aug;54(7):1598-604. doi: https://doi.org/10.1111/and.14425
13. Saleh R, Majzoub A, Abu El-Hamd M. An update on the treatment of premature ejaculation: A systematic review. Arab J Urol. 2021 Aug 4;19(3):281-302. doi: https://doi.org/10.1080/2090598X.2021.1943273
14. Vieiralves RR, Favorito LA. Dapoxetine and premature ejaculation. Int Braz J Urol. 2023 Jul-Aug;49(4):511-4.
doi: https://doi.org/10.1590/S1677-5538.IBJU.2023.9908
15. Tuken M, Culha MG, Serefoglu EC. Efficacy and safety of dapoxetine/sildenafil combination tablets in the treatment of men with premature ejaculation and concomitant erectile dysfunction-DAP-SPEED Study. Int J Impot Res. 2019 Mar;31(2):92-6. doi: https://doi.org/10.1038/s41443-019-0122-2
16. Li J, Liu D, Wu J, Fan X, Dong Q. Dapoxetine for the treatment of premature ejaculation: a meta-analysis of randomized controlled trials with trial sequential analysis. Ann Saudi Med. 2018 Sep-Oct;38(5):366-75. doi: https://doi.org/10.5144/0256-4947.2018.366
17. Ratajczak P, Kus K, Zielińska-Przyjemska M, Skórczewska B, Zaprutko T, Kopciuch D, et al. Antistress and antidepressant properties of dapoxetine and vortioxetine. Acta Neurobiologiae Experimentalis. 2020;80(3):217-24. doi: https://doi.org/10.21307/ane-2020-020
18. Kulacaoglu F. Painful ejaculation induced by venlafaxine: a case report. European Psychiatry. 2022;65(S1):S801-S801. doi: https://doi.org/10.1192/j.eurpsy.2022.2071
19. Liu S, Singh N, Ben W, Mol BW. The integrity of seven randomized trials evaluating treatments for premature ejaculation. Andrologia. 2022;54:e14573. doi: https://doi.org/10.1111/and.14573
20. Quintana GR, Ponce FP, Escudero-Pastén J, Santibáñez-Palma JF, Aguayo-Zuñiga CP, Carrasco-Portiño M, et al. Analysis of the International Index of Erectile Function: psychometric evidence and measurement invariance across relationship status and age generations in a Chilean sample. Sexual Medicine. 2024;12(6): qfae084. doi: https://doi.org/10.1093/sexmed/qfae084
21. Neijenhuijs KI, Holtmaat K, Aaronson NK, Holzner B, Terwee CB, Cuijpers P, et al. The International Index of Erectile Function (IIEF)-A Systematic Review of Measurement Properties. J Sex Med. 2019 Jul;16(7):1078-91. doi: https://doi.org/10.1016/j.jsxm.2019.04.010
22. Lins L, Carvalho FM. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016 Oct 4;4:2050312116671725. doi: https://doi.org/10.1177/2050312116671725
23. Horpinchenko ІІ, Vorobets DZ. [Mechanisms of sexual dysfunction development]. Lviv: LNMU; 2013. 388 p. Ukrainian.
24. Bensken WP, Ho VP, Pieracci FM. Basic Introduction to Statistics in Medicine. Part 2: Comparing Data. Surg Infect (Larchmt). 2021;22(6):597-603. doi: https://doi.org/10.1089/sur.2020.430
25. Pagano M, Gauvreau K, Mattie H. Principles of Biostatistics. CRC Press; 2022. 735 p.
26. Lazar A, Gewirtz-Meydan A, Rosenbaum TY. War-Time Stress and Sexual Well-Being in Israel. International Journal of Sexual Health. 2024;36(1):1-14. doi: https://doi.org/10.1080/19317611.2024.2317169
27. Gul M, Bocu K, Serefoglu EC. Current and emerging treatment options for premature ejaculation. Nature reviews. Urology. 2022 Nov;19(11):659-80. doi: https://doi.org/10.1038/s41585-022-00639-5
28. Russo A, Capogrosso P, Ventimiglia E, La Croce G, Boeri L, Montorsi F, et al. Efficacy and safety of dapoxetine in treatment of premature ejaculation: an evidence-based review. Int J Clin Pract. 2016 Sep;70(9):723-33. doi: https://doi.org/10.1111/ijcp.12843
29. Gillman N, Gillman M. Premature Ejaculation: Aetiology and Treatment Strategies. Medical Sciences. 2019;7(11):102. doi: https://doi.org/10.3390/medsci7110102
30. Zhou Z, Yang Z, Cui Y, Zhang X, Wang Y, Zhang Y. The network meta-analysis of “on-demand” and “daily” use of paroxetine in treating men with premature ejaculation from randomized controlled trials. Andrologia. 2022;54: e14388. doi: https://doi.org/10.1111/and14388
31. Cortese F, Costantino MF, Luzi G. Can We Consider Erectile Dysfunction as an Early Markerof Cardiovascular Disease? JACC Adv. 2023;2(4):100384. doi: https://doi.org/10.1016/j.jacadv.2023.100384
32. Chéhensse C, Facchinetti P, Bahrami S, Andrey P, Soler JM, Chrétien F, et al. Human spinal ejaculation generator. Ann Neurol. 2017 Jan;81(1):35-45. doi: https://doi.org/10.1002/ana.24819
33. Chow J, Thompson AJ, Iqbal F, Zaidi W, Syed NI. The antidepressant sertraline reduces synaptic transmission efficacy and synaptogenesis between identified lymnaea neurons. Front Mar Sci. 2020;7:603789. doi: https://doi.org/10.3389/fmars.2020.603789
34. Yuan S, Deban CE. SSRI-Induced Hypersexuality. Am J Psychiatry Resid J. 2021 Mar;16(3):9-12. doi: https://doi.org/10.1176/appi.ajp-rj.2021.160305
35. Vorobets DZ, Fafula RV, Chaplyk VV, Vorobets MZ, Onufrovych OK, Besedina AS, et al. Erectile dysfunction and quality of life of men affected by hostilities in the Russian-Ukrainian war. Regulatory Mechanisms in Biosystems. 2024;15(1):62-6. doi: https://doi.org/10.15421/022409
Key words: nursing staff, burnout professional, predisposing factor, syndrome, sociodemographic factors, work-related factors
Ключові слова: медсестринський персонал, професійне вигорання, фактор схильності, синдром, соціально-демографічні фактори, фактори, пов’язані з роботою
Abstract
This study aims to investigate the prevalence of occupational burnout among nursing professionals at Shkodra Regional Hospital and identify factors contributing to it. Specifically, the objectives include measuring the burnout rate within this population and analysing demographic and occupational factors that affect burnout levels. This cross-sectional, observational and analytical study used a semi-standardised questionnaire distributed to healthcare professionals over 7 months (September 2024 – March 2025) at the Shkodra Regional Hospital, Albania. The Excel file obtained from Microsoft software underwent statistical processing in SPSS version 26.0 software. The Maslach Burnout Inventory test was subjected to an evaluation of its reliability and consistency with the Cronbach alpha test. The alpha value was found to be 0.79. The level of significance adopted was p<0.05. The mean age among 131 nurses was 35.35±10.37, with a minimum to maximum age of 18-64 years old. Almost 38.2% of the participants belong to the 26-35 age group, which is considered to be a relatively young demographic one. Burnout prevalence turned to be relatively high – 68.9% for 95% CI (59.4-75.3), where 22.3% exhibited high levels of emotional exhaustion, 45.4% of cases high depersonalisation and low personal accomplishment domen in 92.3%. The phenomenon of depersonalization, in combination with two other areas, is worthy of consideration, with a p-value was found to be less than 0.05. It is noteworthy that the majority of these workers, 62.6%, are engaged in shift work with a frequency of 6-10 days per month. Burnout syndrome presented a correlation with sociodemographic factors (marital status, and monthly income) as well as some of the work-related factors (shift work system, weekly working hours, days on shift, work experience, etc). The findings of this study give us a relatively high prevalence of burnout syndrome among the participants (68.9%). We recommend rapid intervention with different policies or strategies that should improve the reduction of burnout among nursing staff. In order to facilitate recovery, it is essential to address burnout, recognise signs of excessive physical fatigue, seek support from colleagues and therapists, set boundaries, and prioritise self-care.
Реферат
Поширеність професійного вигорання: перехресний аналіз серед фахівців з догляду за хворими в лікарнях. Белішa (Готі) С., Зекя І., Петрі О., Браті (Кіка) Б., Фреску Е., Алі Е., Края Дж., Коя Дж., Абазай Е. Це дослідження має на меті дослідити поширеність професійного вигорання серед медсестер у Шкодерській регіональній лікарні та визначити фактори, що сприяють цьому. Зокрема, цілі включають вимірювання рівня вигорання серед цієї групи населення та аналіз демографічних та професійних факторів, що впливають на рівень вигорання. У цьому перехресному, обсерваційному та аналітичному дослідженні використовувалася напівстандартизована анкета, розповсюджена серед медичних працівників протягом 7 місяців (вересень 2024 р. – березень 2025 р.) у Шкодерській регіональній лікарні, Албанія. Файл Excel, отриманий з програмного забезпечення Microsoft, пройшов статистичну обробку в програмному забезпеченні SPSS версії 26.0. Тест Маслаха на вигорання був оцінений щодо його надійності та узгодженості з альфа-тестом Кронбаха. Значення альфа становило 0,79. Прийнятий рівень значущості становив p<0,05. Серед 131 медсестри середній вік становив 35,35±10,37 року, з мінімальним та максимальним віком 18-64 роки. Майже 38,2% учасників належать до вікової групи 26-35 років, яка вважається відносно молодою демографічною групою. Поширеність вигорання виявилася відносно високою і становила 68,9% для 95% ДІ (59,4-75,3), де 22,3% демонстрували високий рівень емоційного виснаження, 45,4% випадків – високий рівень деперсоналізації та низький рівень особистих досягнень у 92,3%. Феномен деперсоналізації в поєднанні з двома іншими областями заслуговує на увагу, при цьому значення p менше 0,05. Варто зазначити, що більшість цих працівників, 62,6%, працюють позмінно з частотою 6-10 днів на місяць. Синдром вигорання корелював із соціально-демографічними факторами (сімейний стан та щомісячний дохід), а також деякими факторами, пов'язаними з роботою (система позмінної роботи, тижневий робочий час, кількість днів у зміні, досвід роботи тощо). Результати цього дослідження свідчать про відносно високу поширеність синдрому вигорання серед учасників (68,9%). Ми рекомендуємо швидке втручання з використанням різних політик або стратегій, які повинні знизити вигорання серед медсестер. Для сприяння одужанню важливо боротися з вигоранням, розпізнавати ознаки надмірної фізичної втоми, звертатися за підтримкою до колег і терапевтів, встановлювати межі та надавати пріоритет турботі про себе.
Burnout has not been characterised as a medical diagnostic category; instead it has been recognised as a psychological syndrome. The term “burnout” was first coined in the 1970s in the USA by Freudenberger, 1974 [1] and Maslach, 1976 [2]. However, the most precise definition was subsequently provided by Maslach and Jackson, 1981 [3], who specified it as “a syndrome of emotional exhaustion, depersonalisation and reduced personal accomplishment that can occur among individuals who do “people-work” of some kind”.
Despite its psychological underpinnings, in the long term, it exerts a direct and detrimental effect on the individual's health [4, 5]. The World Health Organisation (WHO) defines “Burnout” as “an occupational phenomenon resulting from the failure to manage prolonged exposure to chronic stressors in the workplace effectively” [6].
This term has been utilised within the occupational medicine area. The manifestation of burnout syndrome is observed to vary in its presentation across diverse professional contexts. The consequences of professional burnout have been observed to encompass a wide spectrum of effects, including individual health concerns and organisational issues. As posited by Belisha et al., 2025, burnout has been identified as a pervasive problem among medical personnel, particularly those engaged in nursing and general practice [7]. The syndrome has been documented to manifest through three main symptoms: emotional exhaustion, depersonalisation, and a diminished sense of personal accomplishment.
These symptoms are predominantly experienced by the majority of nursing staff [8]. The predominant cause of burnout is identified as emotional exhaustion. The condition is characterised by a persistent state of fatigue, insomnia, and overwhelming stress [9]. As proposed by Petersen et al., 2023, nurses who experience emotional exhaustion frequently encounter emotional and burnout severity [10], which can impede their capacity to cope effectively with the demands of their professional duties [10,11]. In a further study by Lwiza & Lugazia, 2022, the prevalence of burnout among healthcare professionals in developed countries was estimated to range from 40% to 55% [12]. Moreover, a report by the World Economic Forum on the prevalence of burnout in different populations resulted in the following figures for specific countries [13]: The data indicates that 57% of the cases are in the UK, 50% in the US, 37% in Spain, and 30% in Germany and France, respectively [13,14].
The following essay will provide a comprehensive overview of the relevant literature on the subject. In the context of the national present, the prevalence of burnout among medical personnel, with a particular emphasis on nursing staff, remains under-researched. The extant literature on the subject is limited to a handful of recent publications [7,8,15]. This study aims to investigate the prevalence of occupational burnout among nursing professionals at Shkodra Regional Hospital and identify factors contributing to it. Specifically, the objectives include measuring the burnout rate within this population and analysing demographic and occupational factors that affect burnout levels.
MATERIALS AND METHODS OF RESEARCH
This cross-sectional, observational and analytical study was conducted over a 7-month period (September 2024 – March 2025) at the Shkodra Regional Hospital using a semi-standardized questionnaire distributed to healthcare professionals (nurses and midwifery staff). In this study, it was initially determined that only nursing staff would be included, with the exclusion of any medical or auxiliary staff. The inclusion criteria are as follows: the study is open to registered nursing staff working full-time in the hospital setting. Additionally, the study is open to any staff, irrespective of gender, aged between 23 and 65 years, and with a minimum of six months' experience in nursing practice. Staff members who are willing to participate in the study and provide consent will be eligible to take part. The exclusion criteria are as follows: the target demographic group for this study comprises nursing students, interns, and nursing staff with less than six months of experience in nursing practice. The sample calculation was conducted utilising Medicalcul software, which is an online resource that specialises in numerical computations within the medical field. The anticipated response rate was estimated to be in excess of 50% of the total nursing population.
The calculation demonstrated that a minimum of 162 participants were required to complete the online questionnaire, ensuring a confidence level of 95% and a true value within a margin of error of ±5% of the measured value. The questionnaire was uploaded to Microsoft Office software, after which the link was disseminated to all nurses via the WhatsApp website, Messenger, Facebook, and personal e-mails. At the commencement of each month for a period of approximately three months, a reminder was dispatched to the participants to complete the questionnaire. Registered nurses who completed the questionnaires also completed and signed the consent forms. In conclusion, the data from 130 nurses, who had completed all the questions as requested by the working group, were analysed.
In order to assess the determinants of excessive burnout severity, data were collected on a range of sociodemographic variables including, but not limited to, age, sex, residence, marital status, number of family members, educational level and family income. In addition, information was collected on the characteristics and work experiences of the nursing staff in order to provide a comprehensive overview of the subject matter. The second questionnaire is the standardized Maslach Burnout Inventory (MBI), which was developed and validated by Maslach & Jackson in 1981 [3]. This questionnaire is regarded by many researchers as the gold standard for measuring and assessing burnout in different professions [16, 17, 18, 19]. Its high reliability and validity are well-documented. The MBI is comprised of three distinct sections. The initial section concerns Emotional Exhaustion (EE), whereby individuals present with symptoms of fatigue in the workplace, chronic fatigue, sleep disturbances, and physical ailments. The second section is dedicated to Depersonalisation (DP), which can be defined as a form of "dehumanisation" that manifests in interpersonal relationships. The third section of the questionnaire is entitled Personal Accomplishment (PA). In this section, individuals experience a diminution in PA, frequently receiving a negative appraisal and perceiving an inability to progress a situation. A high score in the first two sections and a low score in the last section may be indicative of burnout. It is imperative to note that all questions in this questionnaire are formulated as statements pertaining to personal sentiments or dispositions.
The frequency scale is labelled on each point, starting from 1 to 7, with 1=Never; 2=Few times a year; 3=Once a month; 4=Few times a month; 5=Once a week; 6=Few times a week; 7=Everyday. Following the adaptation of the questionnaire into Albanian, the translated version was evaluated by three specialists in the field. The evaluation was then conducted in a pilot study phase. Following the pilot phase and the implementation of minor linguistic modifications, the MBI test was subjected to an evaluation of its reliability and consistency with the Cronbach alpha test. The alpha value was found to be 0.79.
The initial step in commencing the work for this study was to apply to the ethics committee at the University of Medicine, Tirana, for approval. Firstly, all documentation related to the purpose of the study, the methodology to be employed, the expected results and the involvement of health workers was prepared. Subsequently, the documentation was submitted to the University of Medicine’s Ethics Committee in Tirana for the purpose of obtaining approval and authorisation for the study. Following the received ethical approval from the relevant committee, the investigators proceeded to present themselves to the hospital directorate at which the study was to be conducted. The individuals responsible for authorising and facilitating the execution of this study by the medical personnel have taken measures to ensure that the highest standards of research integrity have been adhered to. The working group convened a preliminary meeting with the employees, during which the purpose and objectives of the study were communicated to each participant. It was emphasised that participation in the study was voluntary and that employees were at liberty to withdraw from the study at any time. It was explained to each participant that no incentives or gifts would be offered. Each participant was assured of the maintenance and storage of the data, with confidentiality guaranteed for any data provided by them. The survey was completed within a time frame of over 30 minutes.
The Excel file obtained from Microsoft software underwent statistical processing in IBM Statistics software SPSS, version 26.0 Issue, DT449052. The data were presented as frequency and percentage, as well as mean and standard deviation (SD). The statistical analysis was conducted using a combination of student t-test, linear correlation, and linear regression analysis. In the context of categorical data, the statistical analysis employed was the chi-squared test. The level of significance adopted was p<0.05.
RESULTS AND DISCUSSION
In the context of this cross-sectional and descriptive study, it is important to reiterate the exclusionary criteria previously outlined in the methodology section. Specifically, the study focused exclusively on nursing and midwifery staff, with the former constituting 83.2% of the sample and the latter making up 16.8%. As illustrated in Table 1, the descriptive socio-demographic data of 131 medical personnel participants is presented, with a mean age of 35.35±10.37, a minimum age of 18 years, and a maximum of 64 years. The data indicates that almost 38.2% of the participants belong to the 26-35 age group, which is considered to be a relatively young demographic. The majority of the subjects are female, accounting for 71.8% of the total, and approximately 74.8% of the participants reside in urban areas. It is important to note that 28.2% of the participants in this study reside with family members, including spouses, children, and parents.
Furthermore, 27.5% of the participants live solely with parents, and it is noteworthy that the majority of these families consist of four to six members (58.2%). With respect to familial income, the majority of respondents (48.9%) reported a family income ranging from 60,001 to 100,000 lek (Table 1).
The second section of the questionnaire, which was drafted by the working group, concerns itself with the issue of employment data and the working hours that respondents perform on a daily basis at the Shkodra Regional Hospital. As anticipated, the vast majority of the participants, approximately 97.7%, are engaged in full-time employment at this institution, with a mere 2.3% engaged in part-time or temporary contractual arrangements.
With regard to the work experience of the medical staff (nurses and midwives), it is evident that a significant proportion of the sample, constituting 33.6% of the total, have length of service from one and five years of experience. However, it is noteworthy that the majority of these workers, 62.6%, are engaged in shift work, while only 37.4% adhere to a standard 8-hour workday. In this study, approximately 50% of the participants (49.6%) reported working on the second shift, with a frequency of 6-10 days per month. Meanwhile, 47.3% of the participants indicated that they worked on the third shift, with a frequency of 6-10 times per month.
Furthermore, it was observed that approximately one-third of the participants (29.8%) had been working in shifts for 1-5 years. A mere 9.2% of nurses and midwives have been working in shifts for less than one year, while 6.9% have been working in shifts for more than 20 years (Table 2).
The following figure presents the levels of burnout experienced by nursing staff for each of the MBI test areas. In the context of the "Emotional Exhaustion" area, 22.3% of the nursing staff exhibited high levels of burnout, while 57.7% demonstrated moderate symptoms. Notably, only 20% of the participants exhibited low levels of burnout. In the case of the “Depersonalisation” area, 45.4% of cases resulted in high levels of burnout, while 54.6% resulted in moderate levels. There were no cases of low burnout. With regard to the “Personal Accomplishment” area, 3.1% of subjects have been identified as experiencing high levels of burnout, 4.6% are exhibiting moderate symptoms, and 92.3% demonstrate low levels of burnout (Fig.).
Variables Categories Frequency Percentage Age group (years) ≤25 years old 25 19.1 26-35 years old 50 38.2 36-45 years old 31 23.7 46-55 years old 18 13.7 56-65 years old 7 5.3 Gender Male 37 28.2 Female 94 71.8 Residence Rural area 33 35.2 Urban area 98 74.8 Marital status Relationship 3 2.3 Single 37 28.2 Divorced 1 0.8 Married 89 68 I/e widow 1 0.8 Education level Higher education 129 98.4 Secondary education 2 1.6 How many members are in your family 1-3 members 36 27.6 4-6 members 76 58.2 ≥7 members 19 14.6 Monthly family income level less than 40,000 lek 2 1.6 40,001-60,000 lek 22 16.8 60,001-100,000 lek 64 48.9 more than 100,001 lek 43 32.8
(exact number)
The following table illustrates the correlation statistics of the scores collected for the three MBI areas. As demonstrated in Table 3, a positive correlation between emotional exhaustion and depersonalisation is evident. Furthermore, DP has been found to correlate with both EM and PA, nonetheless, a negative correlation is evident in the latter case. The phenomenon of depersonalization, in conjunction with two other areas, is worthy of consideration. In all cases, the p-value was found to be less than 0.05 (Table 3).
Correlations Emotional Exhaustion Depersonalization Personal Accomplishment Emotional Exhaustion Pearson Correlation 1 0.528** -0.046 Sig. (2-tailed) 0.000 0.601 Depersonalization Pearson Correlation 0.528** 1 -0.395** Sig. (2-tailed) 0.000 0.000 Personal Achievements Pearson Correlation -0.046 -0.395** 1 Sig. (2-tailed) 0.601 0.000 Notes: * – correlation is significant at the 0.05 level (2-tailed); ** – correlation is significant at the 0.01 level (2-tailed).
In the subsequent table, we have presented the correlations between risk factors and the extent of burnout, according to each area. As is apparent from the data, a significant relationship (positive/negative) is observed between one, two or three domens and several factors, including marital status, family monthly income, employment status, profession, work system, hours of work based on the work system, and the number of times the third shift is worked. The issue of staff shortages, necessitating the replacement of other personnel, is one that is frequently encountered. The question of being overloaded at work is also one that is pertinent. The purpose of this study is to ascertain the level of job satisfaction experienced by respondents. The present study seeks to ascertain whether respondents are satisfied with the conditions of their working environment. What is the nature of your relationship with your colleagues? The final question pertains to the question of whether the individual in question has the support of their superiors in their professional endeavours. In all cases, the p-value was found to be less than 0.05 (Table 4).
Factors correlations Emotional Exhaustion Depersonalization Personal Accomplishment Gender Pearson Correlation 0.006 0.108 -0.153 Sig. (2-tailed) 0.944 0.223 0.083 Residence Pearson Correlation -0.034 0.011 0.023 Sig. (2-tailed) 0.703 0.897 0.795 Marital Status Pearson Correlation 0.363 0.486 -0.394 Sig. (2-tailed) 0.003 0.000 0.008 Educatio level Pearson Correlation -0.037 -0.081 0.065 Sig. (2-tailed) 0.674 0.362 0.461 How many members are in your family Pearson Correlation -0.022 0.084 0.075 Sig. (2-tailed) 0.807 0.341 0.394 Family monthly income level Pearson Correlation -0.357 0.305 0.346 Sig. (2-tailed) 0.006 0.025 0.006 Employes status Pearson Correlation -0.068 0.028 -0.185* Sig. (2-tailed) 0.444 0.754 0.035 Profession Pearson Correlation -0.196* -0.171 0.112 Sig. (2-tailed) 0.025 0.052 0.207 Work system Pearson Correlation -0.297 0.274 0.326 Sig. (2-tailed) 0.012 0.045 0.009 The hours you work are based on the work system Pearson Correlation 0.389** 0.302** -0.159 Sig. (2-tailed) 0.005 0.004 0.07 Working time days/hours Pearson Correlation 0.498** 0.245** -0.130 Sig. (2-tailed) 0.000 0.008 0.125 How many times in month are you on second shift Pearson Correlation 0.139 0.089 0.067 Sig. (2-tailed) 0.234 0.234 0.395 How many times are you on the third shift Pearson Correlation 0.321 0.373 0.285 Sig. (2-tailed) 0.008 0.003 0.045 How many years have you been working shifts Pearson Correlation -0.421** -0.328** .345** Sig. (2-tailed) 0.000 0.000 0.001 Do you often face staff shortages where you have to replace other people? Pearson Correlation 0.321** 0.249** -0.002 Sig. (2-tailed) 0.000 0.004 0.983 Are you overloaded at work? Pearson Correlation 0.434** 0.230** -0.104 Sig. (2-tailed) 0.000 0.008 0.241 Have you been absent from work for health reasons Pearson Correlation 0.113 0.108 0.032 Sig. (2-tailed) 0.201 0.223 0.714 How satisfied are you with your job Pearson Correlation -0.414** -0.399** .286** Sig. (2-tailed) 0.000 0.000 .001 Are you satisfied with the conditions of the environment where you work Pearson Correlation -0.281** -0.230** .094 Sig. (2-tailed) 0.001 0.008 0.288 What is your relationship with colleagues Pearson Correlation -0.341** -0.289** 0.364 Sig. (2-tailed) 0.000 0.001 0.003 Do you have the support of your superiors in your work? Pearson Correlation -0.344** -0.389** 0.269 Sig. (2-tailed) 0.000 0.000 0.045 Notes: * – correlation is significant at the 0.05 level (2-tailed); ** – correlation is significant at the 0.01 level (2-tailed).
Burnout, a syndrome that has the potential to affect healthcare systems on a global scale, has been referred to as the "disease of the 21st century" [7]. The issue of excessive burnout severity in our country has not been the focus of significant research interest, resulting in a paucity of analysis and examination of the predisposing factors that contribute to it. The present study examines the prevalence of the aforementioned syndrome and its correlation with socio-demographic and professional variables that contribute to the condition among healthcare personnel (nursing). The prevalence of burnout among 131 nurses was found to be relatively high at 68.9%, with a 95% confidence interval of [59.4 to 75.3]. The mean value of the EE area was found to be 24.59 for 95% CI [19 to 30], while the participants with the highest EE resulted in 22.3%. The mean value of DP resulted in 13.04 for 95% CI [8-16], where 45.4% of the participants appear to have high DP. The mean value for the PA area was found to be 53%, with a 95% confidence interval of [48.5 to 56]. This indicates a prevalence of low PA, with 92.3% of the participants demonstrating this level of physical activity. This value is relatively high, as demonstrated by a study conducted by Heinen et al., which assessed the prevalence of burnout in 10 European countries. This prevalence was on an average of 28% of nursing staff, where the lowest prevalence was observed in employees in the Netherlands with 10% and the highest in employees in England with 42% [20]. Conversely, the prevalence observed in this study was lower than that reported by Mathkour et al., who found a prevalence rate of burnout among healthcare workers in 73.3% of participants [21]. A substantial body of research has identified various sociodemographic factors [e.g., gender, marital status] and work-related factors [e.g., weekly work hours, monthly night shifts, years of experience, sleep quality, depressive symptoms, anxiety symptoms] as significant contributors to the development of high burnout [22, 23, 24]. In relation to factors such as gender, research indicates that women are more likely to experience physical fatigue syndrome than men [25]. The present study found no significant relationship, which is consistent with the findings of another study [12]. The impact of marital relationships and family stability on individual lives has been demonstrated to be a positive one.
Conversely, an unstable relationship has been shown to engender stress, which can in turn compromise work performance and quality of life [26, 27]. This, in turn, has the potential to increase the risk of burnout syndrome. The findings of this study revealed no significant relationship between burnout and marital status, a finding that aligns with the results of a previous study [26, 27]. A number of studies have indicated that the combination of factors, including extended working hours, shift work systems, protracted work hours, work experience, relationships with colleagues and superiors, frequently contributes to elevated levels of stress among employees, who often report a high degree of burnout [24-28]. In the majority of cases, these employees are potentially vulnerable groups susceptible to excessive fatigue.
The findings of the present study are consistent with those of previous studies, for some of the variables such as marital status, family monthly income, profession, work systems, weekly hours at work, working time days/hours, etc. For each of these variables, a strong correlation was identified between one, two or three areas of the MBI questionnaire, with a p value less than 0.05%. It is imperative to acknowledge the limitations of this study when interpreting the findings. Firstly, the number of nursing staff included was minimal. Secondly, the study focuses exclusively on nursing staff, excluding all other medical personnel. This methodological decision is pivotal in enabling a meaningful comparison between the two groups. Thirdly, the decision was taken to utilise the method of data collection through self-reported questionnaires. However, the reliability of this method of completion is questionable, given the observed discrepancy between cases that refer to depersonalisation and those that demonstrate low personal achievement in their work. This discrepancy may introduce a degree of bias into the responses. Notwithstanding the aforementioned limitations, the study successfully achieved its objectives and constitutes a valuable resource as it is the inaugural study to investigate the prevalence of burnout syndrome and its associated factors among healthcare workers in Shkoder.
CONCLUSION
1. This study has determined a relatively high prevalence rate (68.9%) of burnout syndrome within the surveyed population. A significant proportion of the nursing staff exhibited elevated levels of burnout in the Depersonalisation area, with 45.4% demonstrating high Emotional Exhaustion and 24.59% exhibiting low Personal Accomplishment.
2. The present study identified several factors that were found to be significantly associated with burnout syndrome. These factors included sociodemographic factors, such as marital status and monthly income, as well as work-related factors, including shift work system, weekly working hours, days on shift, and work experience.
3. In order to facilitate recovery, it is essential to address burnout, recognise signs of excessive physical fatigue, seek support from colleagues and therapists, set boundaries, and prioritise self-care.
4. In view of the high prevalence of burnout, it is recommended that prompt intervention be made with various policies or strategies that should lead to a reduction in the incidence of burnout among nursing staff.
5. Achieving an equilibrium between opportunities and work performance is a crucial factor in effective stress management, which will reduce burnout and enhancement of quality of life among healthcare workers. In addition to this, a superior standard of patient care is provided.
Contributors:
Belisha (Hoti) S. – conceptualization, methodology, investigation, project administration, writing – review & editing;
Zekja I. – project administration, supervision, methodology, writing – review & editing;
Petri O. – methodology, investigation, data curation, writing – original draft, formal analysis;
Brati (Kika) B. – methodology, formal analysis, investigation, data curation, writing – original draft;
Fresku E. – data curation, formal analysis, supervision, writing – original draft;
Ali E. – supervision, methodology, investigation, writing – review & editing;
Kraja J. – methodology, investigation, formal analysis, writing – review & editing;
Koja Gj. – investigation, formal analysis, writing – original draft;
Abazaj E. – conceptualization, methodology, investigation, project administration, data curation, formal analysis, supervision, writing – original draft; writing – review & editing.
Acknowledgements
It is imperative to express our profound gratitude to all those members of staff whose invaluable contributions were instrumental in the design, implementation and completion of this study. Secondly, gratitude is extended to all nursing staff of Rajonal Hospital of Shkodra, who participated in this study, responding promptly to the invitation and completing the form in an expeditious manner.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
1. Freudenberger HJ. Staff Burn-Out. Journal of Social Issues. 1974;30(1):159-65. doi: https://doi.org/10.1111/j.1540-4560.1974.tb00706.x
2. Maslach C. Burned out. Human Behavior. [Internet]. 1976 [cited 2025 Aug 23];59:16-22. Available from: https://www.researchgate.net/publication/263847499_Burned-Out
3. Maslach C, Jackson SE. The measurement of experienced burnout. Journal of Occupational Psychology. 1981;2(2):99-113. doi: https://doi.org/10.1002/job.4030020205
4. Barragán Martín AB, Molero Jurado MDM, Pérez-Fuentes MDC, Simón Márquez MDM, Sisto M, Gázquez Linares JJ. Published research on burnout in nursing in Spain in the last decade: bibliometric analysis. Healthcare (Basel). 2020 Nov 12;8(4):478. doi: https://doi.org/10.3390/healthcare8040478
5. Nagarajan R, Ramachandran P, Dilipkumar R, Kaur P. Global estimate of burnout among the public health workforce: A systematic review and meta-analysis. Human Resources for Health. 2024;22:30. doi: https://doi.org/10.1186/s12960-024-00917-w
6. World Health Organization. Burn-out an "occupational phenomenon": International Classification of Diseases [Internet]. 2022 [cited 2025 Aug 23]. Available from: https://www.who.int/news/item/28-05-2019-burn-outan-occupational-phenomenon-international-classification-of-diseases
7. Belisha S, Zekja I, Abazaj E, Dibra V. Prevalence and associated risk factors for occupational burnout among healthcare personnel: a systematic review. Albanian Journal of Trauma and Emergency Surgery. 2025;9(2):1852-62. doi: https://doi.org/10.32391/ajtes.v9i2.448
8. Ramasaco L, Abazaj E, Brati B. Mental Health Burden among Healthcare Workers during the COVID-19 Time in Albania. ASEAN Journal of Psychiatry. 2023;24(1);1-13. doi: https://doi.org/10.54615/2231-7805.47289
9. Butler S. Understanding burnout in nurses: Identification and coping strategies. British Journal of Nursing. 2025;34(4):220-4. doi: https://doi.org/10.12968/bjon.2024.0244
10. Petersen J, Wendsche J, Melzer M. Nurses’ emotional exhaustion: Prevalence, psychosocial risk factors and association to sick leave depending on care setting: a quantitative secondary analysis. J Adv Nurs. 2023;79(1):182-93. doi: https://doi.org/10.1111/jan.15471
11. Kelly L. Burnout, compassion fatigue, and secondary trauma in nurses: recognising the occupational phenomenon and personal consequences of caregiving. Crit Care Nurs Q. 2020;43(1):73-80. doi: https://doi.org/10.1097/CNQ.0000000000000293
12. Lwiza AF, Lugazia ER. Burnout and associated factors among healthcare workers in acute care settings at a tertiary teaching hospital in Tanzania: An analytical cross-sectional study. Health science reports. 2023;6:e1256. doi: https://doi.org/10.1002/hsr2.1256
13. World Economic Forum. Wellbeing and Mental Health. What causes us to burnout at work? [Internet]. 2019 Oct 10 [cited 2025 Aug 23]. Available from: https://www.weforum.org/stories/2019/10/burnout-mental-health-pandemic/
14. Woo T, Ho R, Tang A, Tam W. Global prevalence of burnout symptoms among nurses: A systematic review and meta-analysis. J Psychiatr Res. 2020;123:9-20. doi: https://doi.org/10.1016/j.jpsychires.2019.12.015
15. Merkuri L, Paja E, Hoxhallari K. Burnout among nursing staff: an overview of causes, consequences, and management strategies. MEDICUS. 2025;9(1):38-45. doi: https://doi.org/10.58944/ycqo2677
16. Abdo SAM, El-Sallamy RM, El-Sherbiny AAM, Kabbash IA. Burnout among physicians and nursing staff working in the emergency hospital of Tanta University, Egypt. Eastern Mediterranean Health Journal La Revue de Santé de la Méditerranée orientale. 2015;21(12):906-15. doi: https://doi.org/10.26719/2015.21.12.906
17. Suner-Soler R, Grau-Martína A, Flichtentrei D, Prats M, Braga F, Font-Mayolas S, et al. The consequences of burnout syndrome among healthcare professionals in Spain and Spanish speaking Latin American countries. Burnout Res. 2014;1:82-89. doi: https://doi.org/10.1016/j.burn.2014.07.004
18. Chou LP, Li CY, Hu SC. Job stress and burnout in hospital employees: comparisons of different medical professions in a Regional hospital in Taiwan. BMJ Open. 2014;4:e004185.
doi: https://doi.org/10.1136/bmjopen-2013-004185
19. Schuttle N, Toppinen S, Kalimo R, Schaufeli W. The factorial validity of the Maslach Burnout Inventory across oc cupational groups and nations. J Occup Organ Psychol. 2000;73:53-66. doi: https://doi.org/10.1348/096317900166877
20. Heinen MM, van Achterberg T, Schwendimann R, Zander B, Matthews A, Kózka M, et al. Nurses' intention to leave their profession: a cross sectional observational study in 10 European countries. Int J Nurs Stud. 2013;50:174-84. doi: https://10.1016/j.ijnurstu.2012.09.019
21. Mathkour A, Alzahrani AH, Narapureddy BR, Alqahtani FM, Alshehri AM, Althagafi MA, et al. Prevalence and risk factors of burnout among employees at COVID-19 vaccination centers: A cross sectional study. PloS One. 2025;20(5):e0322803. doi: https://doi.org/10.1371/journal.pone.0322803
22. Zhang X, Wang J, Hao Y, Wu K, Jiao M, Liang L, et al. Prevalence and Factors associated with burnout of frontline healthcare workers in fighting against the COVID-19 Pandemic: Evidence from China. Front Psychol. 2021;12:680614. doi: https://doi.org/10.3389/fpsyg.2021.680614
23. Mohamed NA, Mohamed YA, Haji Mohamud RY, Gabow AA. Evaluation of the prevalence and risk factors of burnout syndrome among healthcare workers: A cross-sectional study. World J Psychiatry. 2025;15(2):98496. doi: https://doi.org/10.5498/wjp.v15.i2.98496
24. Lee YL, Dai JW, Li XW, et al. Prevalence and the associated factors of burnout among the critical healthcare professionals during the post-pandemic era: a multi-institutional survey in Taiwan with a systematic review of the Asian literatures. BMC Public Health. 2024;24:3480. doi: https://doi.org/10.1186/s12889-024-21084-6
25. Anbesaw T, Zenebe Y, Abebe M, Tegafaw T. Burnout Syndrome and Associated Factors Among Health Care Professionals Working in Dessie Comprehensive Specialized Hospital, Dessie, Ethiopia. Sage Open. 2023;13(4):1-9. doi: https://doi.org/10.1177/21582440231215147
26. Dechasa DB, Worku T, Baraki N, Merga BT, Asfaw H. Burnout and associated factors among nurses working in public hospitals of Harari region and Dire Dawa administration, eastern Ethiopia. A cross sectional study. PloS One. 2021;16:e0258224. doi: https://doi.org/10.1371/journal.pone.0258224
27. Chirico F, Afolabi AA, Ilesanmi OS, Nucera G, Ferrari Ge, Sacco A, et al. Prevalence, risk factors and prevention of burnout syndrome among healthcare workers: An umbrella review of systematic reviews and meta-analyses. Journal of Health and Social Sciences. 2021;6(4):465-91. doi: https://doi.org/10.19204/2021/prvl3
28. Merces MCD, Coelho JMF, Lua I, Silva DSE, Gomes AMT, Erdmann AL, et al. Prevalence and factors associated with burnout syndrome among primary health care nursing professionals: a cross sectional study. Int J Environ Res Public Health. 2020;17(2):474. doi: https://doi.org/10.3390/ijerph17020474